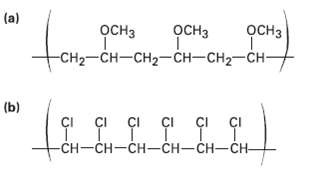
Problem 7.18 Show the monomer units you would use to prepare the following polymers: (a) -CH2-CH-CH2-CH-CH2-CH-
Question:
Problem 7.18 Show the monomer units you would use to prepare the following polymers:
Transcribed Image Text:
(a) дсна -CH2-CH-CH2-CH-CH2-CH- OCH3 осHз осHз (b) çI çI çI çı çI ÇI CI -CH-CH-CH-CH-CH-CH-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
Strategy Find the smallest repeati...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show what reagents you would use to prepare each of the following ethers via a Williamson ether synthesis, and explain your reasoning. a. b. c. OMe
-
Show what reagents you would use to prepare each of the following ethers via an alkoxymercuration-demercuration. a. b. c. d. OEt
-
Identify the reagents you would use to prepare the following compound via a Robinson annulation. . 0=
-
b) A firm produces two types of sugar, A and B at a constant average cost of RM 2 and RM3 per kilogram, respectively. The quantities, q and qg (in kilogram) of A and B that can be sold each week are...
-
A study revealed that the 30-day readmission rate was 31.5 percent for 400 patients who received after-hospital care instructions (e.g., how to take their medications) compared to a readmission rate...
-
Using the given functions, find the limits by plugging in (if possible). Say whether the limit is infinity or negative infinity. Compute the value of the function 0.1 and 0.01 above and below the...
-
Clearly explain the differences among VP, VE, and VA, with an example of each.
-
Journalize the adjusting entry needed on December 31, end of the current accounting period, for each of the following independent cases affecting Rowling Corp. Include an explanation for each entry....
-
Convert the following numbers (Show the steps of your work): a. Ox23734 Binary: Decimal: b. Ob01100011 Hex: Decimal: c. 12345 Hex: Binary:
-
Mini-Case Study: The Back to School Crunch at Global Green Books Publishing Global Green Books Publishing is a successful printing and publishing company. Just two years old, it has taken on a great...
-
Propose structures for alkenes that yield the following products on reaction with ozone followed by treatment with Zn: (a) (CH3)2C = O + H2C = O (b) 2 equiv CH3CH2CH = O
-
One of the chain-termination steps that sometimes occurs to interrupt polymerization is the following reaction between two radicals. Propose a mechanism for the reaction, using fishhook arrows to...
-
The following is a list of accounts and amounts reported for Rollcom, Inc., for the fiscal year ended September 30, 2015. The accounts have normal debit or credit balances. Required: 1. Prepare an...
-
Rank the following three single taxpayers in order of the magnitude of taxable income. Assume none of the taxpayers contributed to charity this year. Note: First mean highest taxable income, third...
-
On 1 0 July 2 0 2 2 Coolum Ltd provides some consulting advice to Florida Inc. ( US ) for an agreed fee of US$ 1 million. The amount is paid into the US bank account of Coolum Ltd on 1 0 July 2 0 2 2...
-
Following are the transactions of a new company called Pose-for-Pics. August 1 M. Harris, the owner, invested $10,500 cash and $45,150 of photography equipment in the company in exchange for common...
-
For the past year, a firm had sales of $41,987, interest expense of $3,232, cost of goods sold of $16,500, selling and administrative expense of $11,000, and depreciation of $6,200. If the tax rate...
-
Manufacturers Southern leased high - tech electronic equipment from International Machines on January 1 , 2 0 2 4 . International Machines manufactured the equipment at a cost of $ 9 5 , 0 0 0 ....
-
What is the difference between body waves and surface waves? Which of these seismic waves reveals key information about Earths internal structure?
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
Use Figure 12.28 to estimate the boiling point of water at an external pressure of 200 torr. (a) 66 C (b) 84 C (c) 100 C (d) 0 C Vapor pressure (torr) 800- 760 600- 400- 200 0- 0 34.6 C Diethyl ether...
-
Arrange the compounds within each set in order of increasing acidity (decreasing pK") in solution. Explain your reasoning. (a) 2-chloro- 1-propanethiol, 2-chloroethanol, 3-chloro- 1-propanethiol (b)...
-
Normally, dibutyl ether is much more soluble in benzene than it is in water. Explain why this ether can be extracted from benzene into water if the aqueous solution contains moderately concentrated...
-
Nonactin (structure on p. 354) forms a strong complex with the ammonium ion, +NH4. What types of interactions are expected between the nonactin molecule and the bound ion? Contrast these interactions...
-
Compare and contrast digital evidence and non-digital evidence. Provide three examples of how they are similar and three examples of how they are different. You must use the provided course materials...
-
In May of 2015, Mr. Miradi made an oral agreement with Mr. Miondoko for the sale of a piece of land situate in Nyeri County Land title No. Gakawa/Kahurura Block III/Mwichwiri/823 for a price of KShs....
-
and QB- Two identical point charges (+2.27 10-9 C) are fixed in place, separated by 0.460 m (see the figure). Find (a) the electric field and (b) the electric potential at the midpoint of the line...

Study smarter with the SolutionInn App


