Propose a synthesis of the drug sulfathiazole from benzene and any necessaryamine. Sulfathiazole H2N
Question:
Propose a synthesis of the drug sulfathiazole from benzene and any necessaryamine.
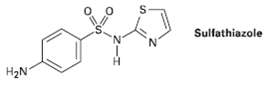
Transcribed Image Text:
Sulfathiazole H2N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
This reaction sequence is similar to the sequence used to synthesize sulfanilamide Key s...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a synthesis of 4, 4-dimethyl-2-pentyne (the compound in Problem 14.19) from an alkyl halide and an alkyne.
-
Propose a synthesis of the anti-inflammatory drug Fenclorac fromphenyl-cyclohexane. CI Fenclorac CHCO2H
-
Propose a synthesis of (R)-CH3CHN3CH2CH3, starting from (R)-2-chlorobutane.
-
Match the following ratios with the appropriate formula. Ratio or Rate Formula a. Income from operations Interest expense Acid-test Total liabilities Stockholders' equity Current b. Net income-...
-
Do you agree with how Adam Silver handled the situation? Do leaders really set the tone of an organization's values?
-
When pseudoionone is treated with BF3 in acetic acid, ring closure takes place and α-and β-ionone are produced. This is the next step in the vitamin A synthesis. (a) Write...
-
A nonstatistical sampling plan can: a. Overstate the estimate of sampling risk. b. Misdirect an auditor to unreliable sampling units. c. Replicate the results of a statistical sampling plan. d....
-
Vodra was employed as a salesperson and contracting agent for American Security Services. As part of his contract of employment, Vodra signed an agreement that for three years after leaving this...
-
A tuning fork is sounded above a resonating tube (one end closed), which resonates at a length of 0.20 m and again at 0.60 m. If the tube length were extended further, at what point will the tuning...
-
1. The endpoints of the bottom side of a parallelogram are at A(2, 1) and B(10, 7). The other two sides both have slope 3, and length 5 cm. (a) (2 points) Provide a very neat and accurate sketch this...
-
What products would you expect from Hofmann elimination of the following amines? If more than one product is formed, indicate which ismajor. NH2 (b) NH2 (a) CH3CH2CH2CHCH2CH2CH2CH3 NHCH2CH3 (d) NH2...
-
Propose syntheses of the following compounds from benzene: (a) N, N-Dimethylaniline (b) p-Chloroaniline (c) m-Chloroaniline (d) 2, 4-Dimethylaniline
-
Give three explanations for the law of demand
-
The skier departs the horizontal ski run with a speed of v=110 km/h, soaring into the air. Upon landing, the slope he/she encounters forms a 45 angle with the horizontal plane. Calculate the skier's...
-
A missile is fired off the surface of the Earth with speed v at a 45 degree angle. It reaches a maximum height above the surface of 6,370 km (which is coincidentally equal to the radius of the...
-
Steve Nash makes a free-throw shot at the basket. The mass of the basketball is 0.62 kg. The basketball leaves his hand at a speed of 7.2 m/s from a height of 2.21 m above the floor. Using a...
-
Two students push on a crate to slide it across the floor. One student exerts a force of 7 N to the east, and the other student exerts a force of 4 N to the south. What is the magnitude of the net...
-
10) An ice cube of mass 0.033 kg and temperature -9 C is heated until it is now fully melted and at a temperature of 11C now. What percentage of the total energy was used to melt the ice? (Assume...
-
Why is it important to evaluate increases and decreases in operating expenses? (a) Increases in operating expenses may indicate inefficiencies, and decreases in operating expenses may be detrimental...
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
At 70 K, CCl 4 decomposes to carbon and chlorine. The K p for the decomposition is 0.76. Find the starting pressure of CCl 4 at this temperature that will produce a total pressure of 1.0 atm at...
-
Divide the following compounds into groups that might be expected to exhibit similar chemical behavior: a. C4H10 b. CH3OCH3 c. C3H7OH d. C8H18 e. HOCH2CH2CH2OH f. CH3NH2 g. CH3CH2CH3 h. CH3OH i....
-
Write an equation similar to eq. 1.2 for the formation of a fluorine molecule from two fluorine atoms.
-
Using Table 1.6, write a structural formula for each of the following: a. An alcohol, C3H8O b. An ether, C4H10O c. An aldehyde, C3H6O d. A ketone, C3H6O e. A carboxylic acid, C3H6O2 f. An ester,...
-
Explain the concept of hybrid thread models in operating systems. How do they attempt to combine the benefits of user-space and kernel-space threading ?
-
Describe the characteristics of the six stages of development for intimacy in a romantic relationship using a real or hypothetical example to illustrate the general developmental path.
-
Consider the following payoff matrix: Player 1 Strategy B Strategy A Strategy A (4,3) (6,2) Player 2 Strategy B (3,5) (2,4) Is the payoff matrix symmetric (Yes or No)?

Study smarter with the SolutionInn App


