Propose structures for amines with the following 1H NMR spectra: (a) C3H9NO (b)C4H11NO2 TMS O ppm 10
Question:
Propose structures for amines with the following 1H NMR spectra:
(a) C3H9NO
(b)C4H11NO2
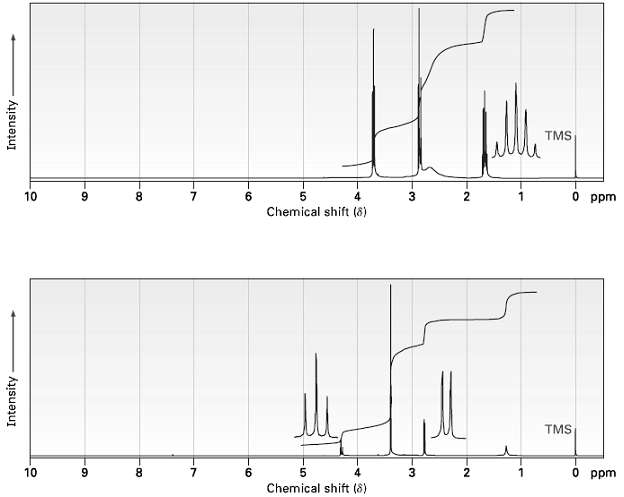
Transcribed Image Text:
TMS O ppm 10 6. Chemical shift (8) TMS O ppm 10 7. 9. Chemical shift (8) Intensity Intensity -3-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a HOCHCHCHNH a 17...View the full answer

Answered By

Robert Mwendwa Nzinga
I am a professional accountant with diverse skills in different fields. I am a great academic writer and article writer. I also possess skills in website development and app development. I have over the years amassed skills in project writing, business planning, human resource administration and tutoring in all business related courses.
4.90+
187+ Reviews
378+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for compounds with the following 1H NMR spectra: (a) C 5 H 9 ClO 2 ? ?IR: 1735 cm ?1 ? (b) C 7 H 12 O 4 ? ? ?IR: 1735 cm ?1 ? (c) C 11 H 12 O 2 ? ??IR: 1710 cm ?1 ? TMS 8. 6. O ppm...
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
A random variable follows the continuous uniform distribution between 60 and 95. a. Calculate the following probabilities for the distribution: 1. P(x > 63) 2. P(x > 70) 3. P(x > 88) 4. P(x = 75) b....
-
Knowing the jargon and "hot button" issues in a particular profession or industry can give you a big advantage when it comes to writing your résumé and participating in job interviews....
-
Barbituric acid dissociates as follows: (a) Calculate the pH and fraction of dissociation of 102.00 M barbituric acid. (b) Calculate the pH and fraction of dissociation of 1010.00 M barbituric acid....
-
In reporting on compliance with laws and regulations, what does the GAO's Government Auditing Standards require an auditor to report?
-
Wainwright Corporation had the following activities in 2010. 1. Sale of land.............................................................$180,000 2. Purchase of...
-
From the reading, there are several things you need to assess and complete for your final project. They are the following: Give an overview of the article and what it says about diversity in law...
-
assembly language ; 6. Temperature conversion between Celsius and Fahrenheit can be computed using ; ; ; the relationship C = (5 / 9) * (F - 32) where C and F are in degrees (signed integer). Celsius...
-
The antitumor antibiotic mitomycin C functions by forming cross-links in DNA chains. (a) The first step is loss of methoxide and formation of an iminium ion intermediate that is deprotonated to give...
-
Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
-
Two plane mirrors intersect at right angles. A laser beam strikes the first of them at a point 11.5cm from their point of inter-section, as shown in Fig. 3338 for what angle of incidence at the first...
-
How do you write this in python? 1 y = I +++ Please help me understand how to do so. Thank you
-
Tax credits generated by a limited partnership benefit the limited partners by A) reducing tax liability dollar for dollar.. B) adding to the appreciation of the real estate properties. C) reducing...
-
Margaret Thatcher uses gas to heat her home and has collected data on her monthly gas bill and monthly heating degree-days. The heating degree-days for a month are calculated by subtracting 65 from...
-
1. Strongest Motivations for Fashion to Acquire Flavoring : Fashion Inc. has several compelling reasons to consider acquiring Flavoring International: Earnings Growth : Flavorings earnings growth...
-
Question. identify the generally accepted accounting conception or constraint violated in the following situation. GuardIt Ltd., provides security services to other businesses.GuardIt Ltd. charges...
-
Exhibits 2.4 and 2.5 in the textbook show industry averages for inventories and net fixed assets as a percentage of total assets taken from The Risk Management Association, Annual Statement Studies....
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Define pOH. What pOH range is considered acidic? Basic? Neutral?
-
Write structural formulas for the following compounds: a. P-chlorostyrene b. 2,3,5-trifluoroaniline c. O-chlorophenol d. Allylbenzene e. P-isopropylphenol f. P-dimethylbenzene g. 1,3-diphenylhexane...
-
Name the following compounds: a. b. C. CI C Br d. Cl e. f. CH3 NO2 CH3 Cl g- h. CH CH, Br Br
-
Give the structures and names for all possible a. Trimethylbenzenes b. Dibromobenzoic acids c. Dinitroanisoles d. Dichloronitrobenzenes
-
Image transcription text Introduction: The Smart Parking Management System (SPMS) project aimed to develop an efficient, automated solution for managing parking spaces in urban areas using Arduino...
-
A. Can we use human capital theory to explain "Age-Discrimination"? If so, how? B. Why do some firms prefer to hire younger workers, especially for entry level positions? C. Why do some firms...
-
Image transcription text Question 6 Not yet answered Marked out of 1.00 '1" Flag question During a mountain?biking tripr Bill and Barry are discussing the merits of having the springs and shocks used...

Study smarter with the SolutionInn App


