Propose structures for compounds with the following 1H NMR spectra: (a) C 5 H 9 ClO 2
Question:
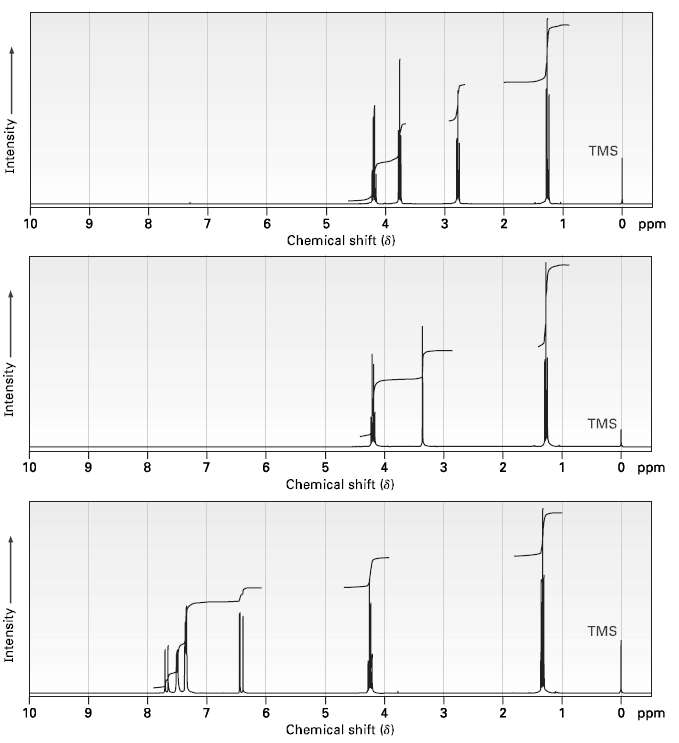
Propose structures for compounds with the following 1H NMR spectra:
(a) C5H9ClO2? ?IR: 1735 cm?1?
(b) C7H12O4? ? ?IR: 1735 cm?1?
(c) C11H12O2? ??IR: 1710 cm?1?
Transcribed Image Text:
TMS 8. 6. O ppm 10 9. 7. Chemical shift (8) TMS O ppm 10 8. 6. Chemical shift (8) TMS O ppm 10 8. 6. Chemical shift (8) Intensity Intensity Intensity 3. 3. 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (18 reviews)
a CICHCHCOCH2CH3 b a 138 b 28 ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
Propose structures for compounds X, Y, and Z: The 1H NMR spectrum of X gives two signals, a multiplet at δ 7.3 (5H) and a singlet at δ 4.25 (2H); the 680-840-cm-1 region of...
-
Propose structures for compounds E and F. Compound E (C8H6) reacts with 2 molar equivalents of bromine to form F (C8H6Br4). E has the IR spectrum shown in Fig. 9.50. What are the structures of E and...
-
Chuck, a single taxpayer, earns $86,000 in taxable income and $20,000 in interest from an investment in City of Heflin bonds. (Use the U.S. tax rate schedule.) Required: a. If Chuck earns an...
-
What are primary and secondary data, and why is it best to start your research by gathering secondary data first?
-
Specify the following additional queries on the database of Figure 3.5 in SQL. Show the query results if applied to the database of Figure 3.6. (a) For each department whose average employee salary...
-
The numbers of stolen bases during the 2016 season for Chicago Cubs players who stole at least one base are listed. 2 8 3 2 13 12 6 2 1 5 11 1 Determine whether the data are qualitative or...
-
Lance H. and Wanda B. Dean are married and live at 431 Yucca Drive, Santa Fe, NM 87501. Lance works for the convention bureau of the local Chamber of Commerce, while Wanda is employed part-time as a...
-
A block of 185F iron falls into an insulated container which contains 0.8ft 3 of 70F liquid water. At the same time the water is stirred by a blade attached to a 200W motor. After 10 minutes the...
-
How can Chi-square analysis be used to distinguish between linkage and independent assortment?
-
Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra: 100 80 60 40 20 4000 3000 2500 2000 1000 500 3500 1500 Wavenumber (cm-1) TMS O ppm 10 6. Chemical shift (8)...
-
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H 2...
-
Even when the sample conditional distributions in a contingency table are only slightly different, when the sample size is very large it is possible to have a large X 2 statistic and a very small...
-
In this Java code, which of these class members are inherited and accessible in the subclass? public class Super { public int x; protected String; private double y; public abstract void doStuff();...
-
How are binary search trees useful in programming? Give three examples of applications/problems that can be helped by using BSTs. b. Question 2: How are heaps useful in programming? Give three...
-
Differences arising between the actual and expected values of the pension contributions What are actuarial losses or gains in a defined benefit plan?
-
What would be a good closing paragraph to end the paper concerning Targets net liabilities and net tangible assets?
-
A H NMR spectrum is shown for a molecule with the molecular formula of C5H10O2. Draw the structure that best fits this data. M 10 9 8 6 5 4 Select to Edit 3 2 Prin
-
An abortion rights advocate wants to estimate the percentage of people who favour opening abortion clinics. She conducts a nationwide survey of 1680 randomly selected adults 18 years and older. The...
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
Name the hybridization scheme that corresponds to each electron geometry. a. Linear b. Trigonal planar c. Tetrahedral d. Trigonal bipyramidal e. Octahedral
-
Which should be faster: bromination of benzene or bromination of N.N-dimethvlaniline? Explain your answer carefully. N,N-dimethylaniline
-
Predict the predominant product(s) from: Mononitration of m-bromoiodobenzene
-
In each of the following sets, rank the compounds in order of increasing harshness of the reaction conditions required to accomplish the indicated reaction. Friedel-Crafts acylation of chlorobenzene,...
-
A proton (mass of 1.67 x 10-27 kg) is moving at 1.25 x 106 m/s directly toward a stationary helium nucleus (mass 6.64 x 10-27 kg). After a head-on elastic collision, what is the velocity of the...
-
The December 31, Year 4, balance sheet for Vernon Corporation is presented here. These are the only accounts on Vernon's balan sheet. Amounts indicated by question marks (?) can be calculated using...
-
Reyes Manufacturing Company uses a job order cost system. At the beginning of January, the company had one job in process (Job 201) and one job completed but not yet sold (Job 200). Job 202 was...

Study smarter with the SolutionInn App


