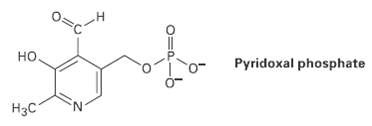
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions.
Question:
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom.
Transcribed Image Text:
но. Pyridoxal phosphate Нас 0=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
sp3 sp2 H3C H sp2 sp2 sp2 sp2 5p3 Pyridoxa...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the molecular structure and the bond angles for each molecule or ion in Exercises 57, 58, and 60.
-
Predict the molecular structure and the bond angles for each of the following. a. SeO3 b. SeO2 c. PCl3 d. SCl2 e. SiF4
-
Predict the molecular structure and the bond angles for each of the following. (See Exercises 89 and 90.) a. XeCl2 b. ICl3 c. TeF4 d. PCl5
-
In the Akerlof example, the individuals are treated as indifferent to risk. What would you expect to see in these markets if individuals wanted to avoid risk? What if there were some risk lovers?
-
Glen and Diane Okumura (both age 48) are married, file a joint return, and live at 39 Kaloa Street, Honolulu, HI 96815. Glen's Social Security number is 111-11-1111, and Diane's is 123-45-6789. The...
-
Each year Forbes reports on the value of all teams in the National Football League. Although England's soccer team, Manchester United, is the most valuable team in the world ($1.8 billion), the NFL...
-
For the system in Problem 6.7, how many hours of sunlight are needed to ensure that the battery bank is at \(100 \%\) charge at the end of the day assuming the same load? Problem 6.7 A PV battery...
-
W. W. Phillips Company produced 4,000 leather recliners during the year. These recliners sell for $400 each. Phillips had 500 recliners in finished goods inventory at the beginning of the year. At...
-
Martin Clothing Company is a retail company that sells hiking and other outdoor gear specially made for the desert heat. It sells to individuals as well as local companies that coordinate adventure...
-
On January 1, 20X1, Pluto Company acquired all of Saturn Companys common stock for $1,000,000 cash. On that date, Saturn had retained earnings of $200,000 and common stock of $600,000. The book...
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
Why do you suppose no one has ever been able to make cyclopentyne as a stable molecule? Cyclopentyne
-
Identify the primary difference between a fault and a no fault ground for divorce.
-
Evaluate f(x) = 5000 (2) for x = -1.5 Round to the nearest hundredths
-
Factor ( 9 + h ) ^ 3 - 7 2 9
-
Suppose a pendulum is Z meters long. The time, t, in seconds that it takes to swing back and forth once is given by t = 2.01 L. If a pendulum is 9.61 meters long, how long does it take to swing back...
-
Rewrite as a logarithmic equation. 1 0 ^ y = 3
-
Factor completely. 8x3 + 27y3
-
What is a multiple bar graph, and how is it helpful?
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Can a fluid obeying the virial equation of state have a vapor-liquid transition?
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
A car is traveling around a banked curve with a radius of 100 meters and a banking angle of 30 degrees at a speed of 40 m/s. What is the coefficient of friction between the tires and the road that...
-
A spring has a force function of F(x) = 3x, where x is the displacement from the equilibrium position. If an object with a mass of 2 kg is moved from x = 0 to x = 4 meters, how much work is done by...
-
A crane lifts a load with a force of 1000 N over a distance of 10 meters in 20 seconds. What is the average power generated by the crane?

Study smarter with the SolutionInn App


