Show equations for the major fragmentations you would expect from the molecular ions of these compounds. List
Question:
Show equations for the major fragmentations you would expect from the molecular ions of these compounds. List the m/z of the productions.
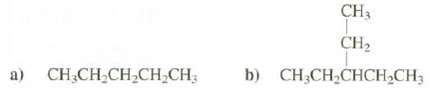
Transcribed Image Text:
CH3 CH2 a) CH;CH-CH,CH-CH; b) CH;CH,CHCH2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a CHCHCHCHCH CH3 CH b CH3C...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What are the major products you would expect from Hofmann elimination of the following amines?
-
Draw the products you would expect from conrotatory and disrotatory Cyclizations of (2Z, 4Z, 6Z)-2, 4, 6-octatrienc. Which of the Iwo paths would you expect the thermal reaction to follow?
-
Formulate the product(s) that you would expect from each of the following reactions. Show stereochemistry clearly. Ch (b) trans-3-Heptene HCI Br2, H2O (a) (c) 1-Ethylcyclohexene CH,CH3 CH3 NaOH, H,O...
-
A house girl added 20g of sodium chloride (NaCl) to 80g of water (atomic masses are Na=23amu, Cl=35.5amu). Calculate a)Percent(w/w) of NaCl b)Mole fraction of NaCl
-
Describe the budgeting process for your selected project. Was bottom-up, top-down, or program budgeting used? Who was involved in the budgeting process? How were costs estimated? What steps are in...
-
List and describe five factors that must be considered with an investment in an overseas manufacturing entity?
-
Using the approach described in Example 2.6, derive the expressions for all the averaged stiffnesses for the planar isotropic lamina in terms of invariants. Use these results to find the...
-
H.J. Heinz Company uses standards to control its materials costs. Assume that a batch of ketchup (6,000 pounds) has the following standards: The actual materials in a batch may vary from the standard...
-
Based on an aging of accounts receivables, management assigned 1% to the $100,000 of receivables 0-30 days outstanding, 5% to the $10,000 receivables 31-60 days and 20% to the $1,000 of receivables...
-
Consider the following 0x86 program: .data array DWORD 1,2,3,4,5,6,7,8,9 aravSize -(S-array)/4 ; array .code main PROC mov ecx,arraySize-1 mov esi.OFFSET array L1: mov eax. [esi] sda mov bx,2 idiv bx...
-
Show the molecular ions formed from these compounds: b) a) CH,NHCH,
-
(a) The base ion in the mass spectrum of 3-ethyl-2-methylpentane occurs at m/z 43m; show the fragmentation that produces this ion. (b) What other fragment would you predict to provide a major peak in...
-
Firms A and B are each considering an unanticipated new investment opportunity that will marginally increase the value of the firm and will also increase the firm's level of diversification. Firm A...
-
The grapevine is a supplement in an organization where formal communication does not work. The grapevine creates a sense of unity among the employees who share and discuss their views with each...
-
Why should you not wait until project completion to perform the Close Project activities? Also, list at least five pitfalls to avoid. Using The Wall Street Journal or other scholarly source, research...
-
Why is entropy as defined for discrete random variables not very useful for continuous random variables? What is quantization and why must there always be a trade-off between quantization accuracy...
-
Write an essay of 1,250-1,500 words that thoroughly explores the following: Many states have adopted three-strikes laws and some have two-strikes laws that often result in life sentences for...
-
Why was it so important that Mr. Sharma restrict his water intake when he went home? What would happen if he did not limit his water intake? If Mr. Sharma found water restriction too difficult, his...
-
What is a genetic cross?
-
Selected condensed data taken from a recent statement of financial position of Morino Ltd. are as follows. MORINO LTD. Statement of Financial Position (partial) Other current assets...
-
Failure to abide by the rule of law makes the return on capital more dependent on the whims of individuals than on laws. Property rights become less secure, which discourages businesses from...
-
The compounds in each part below have the same (or similar) molecular weights. Which compound in each part would you expect to have the higher boiling point? Explain your answers. (a) (b) (c) OH or...
-
Arrange the following compounds in order of increasing boiling point. Explain your answer in terms of the intermolecular forces in each compound. (a) (b) (c) (d)
-
Use arguments based on resonance and electronegativity effects to explain the trend in carbonyl IR stretching frequencies from higher frequency for esters and carboxylic acids to lower frequencies...
-
ABC Hospital is one of two hospitals in the community. The Hospital offers all major specialty services (e.g. cardiology, neurology, pulmonology, etc.) The Hospital is approached by a cardiologist...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $27,760 of office salaries and $70,840 of sales salaries. Withholdings from the employees' salaries...

Study smarter with the SolutionInn App


