Show syntheses of these compounds from1-bromobutane: b) TH. c)
Question:
Show syntheses of these compounds from1-bromobutane:
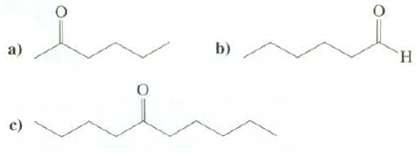
Transcribed Image Text:
b) TH. c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a b h HO HSO4 HgSO4 ...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indication starting materials. Your syntheses may produce both enantiomers of any target that ischiral. a) CH.CH,CH,CH,CH,OCH, from CH;CH,CH,CH=CH2 CH3 b)...
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Show syntheses of these compounds from the indicated startingmaterials. a) CH,CH,CH,CH,N(CH3), from compounds with none of the CN bonds of the final product. CH3 b) CH,CH,OCHCH,CH; from alkyl halides...
-
Consider the following molecule: Tin II Chloride(SnCl2) Are the bonds polar or non-polar? Is the molecule polar or non-polar?
-
Define Web 2.0.
-
The L-shaped object in Figure 11-40 consists of three masses connected by light rods. Find the work that must be done on this object to accelerate it from rest to an angular speed of 2.35 rad/s about...
-
Consider a perpetual American put option (with \(T=\infty\) ). For small stock prices it will be advantageous to exercise the put. Let \(G\) be the largest such stock price. The time-independent...
-
Assume Sally is a professor who has a PhD in accounting and teaches tax courses. She decides that it would help her teaching to earn a law degree with a heavy emphasis on tax classes. Can she deduct...
-
In Marigold Company, the Cutting Department had beginning work in process Inventory of 4000 units, completed and transferred out 26000 units, and fiad an ending work in process inventory of 3500...
-
Do you think employers must choose between agreeable employees and top performers? Why or why not?
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
Show the products of thesereactions: CH CH;CH C3D CH CH3 .. -- NaOH CH3
-
How might Brown have phrased its covenant not to compete with Johnson in such a way that it would have been enforced?
-
Snell's law helps us to determine How much time light spends in a material The angle by which light bends when entering a different material The speed of light in a vacuum with standard temperature...
-
A cannon with its open end at an initial height of 1.5 meters above the ground fires a cannonball at an angle of 40 above the horizontal. It travels a horizontal distance of 40.0 meters to a 4.0...
-
A lead block is suspended from your hand by a string. The force of gravity on the block is balanced by the force exerted by the O block on the Earth O block on the string O string on the block O...
-
The speed of light is constant regardless of the material O True O False
-
If one measures the index of refraction to be 1.1, this material would be More dense than vacuum More dense than acrylic More dense than water O Less dense than air
-
Develop and solve a future-value, a present-value, a future value of an annuity, and a present value of an annuity problem. Establish the three known variables in each problem and solve for the...
-
What are the 5 Cs of marketing channel structure?
-
Instead of the titration of a strong acid by a strong base considered in Question 5, consider the titration of a strong base by a strong acid. Compare and contrast a strong acidstrong base titration...
-
Propose a structure for the product with formula C9H17N that results when 2-(2-cyanoethyl) Cyclohexanone is reducedcatalytically. CH2CH2CN H/Pt C9H17N
-
Coniine, C8H17N, is the toxic principle of the poison hemlock drunk by Socrates. When subjected to Hofmann elimination, coniine yields 5-(N, N-dimethylamino)-1-octene. If coniine is a secondary...
-
How would von synthesize coniine (Problem 24.61) from acrylonitrile (H2C = CHCN) and ethyl 3-oxohexanoate (CH3CH2CH2COCH2CO2Et)?
-
A couple obtained a $20,000 mortgage loan at an interest rate of 10.5% compounded monthly. (Original principal equals to PV of all payments discounted at the interest rate on the loan contract) (1)...
-
What strategies and tactics are employed to manage strategic risks and uncertainties, including geopolitical instability, supply chain disruptions, and emerging competitive threats, while preserving...
-
How do strategic planners integrate ethical considerations and sustainability imperatives into strategic planning processes, balancing short-term financial objectives with long-term societal and...
Effective C An Introduction To Professional C Programming 1st Edition - ISBN: 1718501048 - Free Book

Study smarter with the SolutionInn App


