Show the conjugate acid of each of these species: a) NH d) CHC=C: b) :0: -N: c)
Question:
Show the conjugate acid of each of these species:
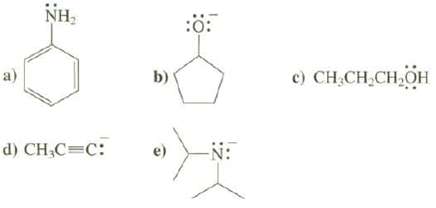
Transcribed Image Text:
a) NH₂ d) CH₂C=C: b) :0: -N: c) CH₂CH₂CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
To get the conjugate acid ...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
a. Draw the conjugate acid of each of the following: (1) NH3 (2) Cl- (3) HO- (4) H2O b. Draw the conjugate base of each of the following: (1) NH3 (2) HBr (3) HNO3 (4) H2O
-
What is the conjugate acid of each of the following bases? (a) HSO4- (b) H2O (c) CH3NH2 (d) -NH2 (e) CH3CH2 (f) CH3CO2-
-
Write the formula for the conjugate acid of each of the following bases: (a) HS-, (b) HCO3-, (c) CO32-, (d) H2PO4-, (e) HPO42-, (f) PO43-, (g) HSO4-, (h) SO42-, (i) SO32-.
-
The labor force participation rate is increasing in Japan because... a) there are fewer unemployed people b) the working age population is increasing c) retirement ages are increasing d) more women...
-
Green Ltd produces a special valve used in the burners of gas stoves. The firm uses the first-in, first-out (FIFO) process costing method for product costing. The costs entered into work in process...
-
Suppose that the price of a commodity is $3.50 in the United States and 4 in the European Monetary Union and the actual exchange rate between the dollar and the euro is R = $1/1, but, the equilibrium...
-
As shown in V9.25 and Fig. P9.105, a spoiler (i.e., an upside-down airfoil) is mounted above the rear wheels of a race car to produce negative lift (i.e., downforce), thereby improving tractive...
-
With 19 different lodging or extended stay brands under the Marriott corporate umbrella, is Marriott at risk of making any positioning errors discussed in the chapter? That is, do they have too many...
-
Explain why the price elasticity of demand determines the incidence of a commodity tax levied on the supplier? Explain why a tax on alcohol may have a deleterious impact on the families of alcohol...
-
Electronic Heaven, Inc., sells electronic merchandise, including a personal computer offered for the first time in September, which retails for $695. Sales of this personal computer for the next...
-
Explain whether each of the following solvents would be acceptable for reactions involving this anion: (a) Liquid NH3 (b) CH3CH2OH (c)CH3CH2OCH2CH3 CHC=C: solvent
-
Show the most stable conjugate base of these compounds: a) CHCHOH i b) HOCCHCHOH c) HNCHCHOH
-
Let A S n + be a symmetric, positive semi-definite matrix. 1. Show that the trace, trace A, and the Frobenius norm,A F, de-pend only its eigenvalues, and express both in terms of the vector of...
-
Describe what sexual harassment is and what forms it can take.
-
Given the Irwin-Hall distribution with pdf, \[f_{Y}(y)=\frac{1}{2(n-1) !} \sum_{k=0}^{n}(-1)^{k}\left(\begin{array}{l}n \\k\end{array} ight)(y-k)^{n-1} \operatorname{sign}(y-k)\] where the sign...
-
Describe what is meant by retaliatory and constructive discharge and how to defend a claim for unfair discharge.
-
Discuss under what conditions unemployment compensation can be denied to a claimant.
-
Explain public policies that protect employees from unlawful discharge.
-
Explain what is meant by a steady state. In the Solow model, which variables are constant in a steady state?
-
Refrigerant R-12 at 30C, 0.75 MPa enters a steady flow device and exits at 30C, 100 kPa. Assume the process is isothermal and reversible. Find the change in availability of the refrigerant.
-
Evaluate (2i + 2'). i=1
-
Where would you expect each of the following compounds to absorb in the IR spectrum? (a) 4-Penten-2-one (b) 3-Penten-2-one (c) 2, 2-Dimethylcyclopentanone (d) m-Chloro benzaldehyde (e)...
-
How might you use mass spectrometry to distinguish between the following pairs of isomers? (a) 3-Methyl-2-hexanone and 4-methyl-2-hexanone (b) 3-Heptanone and 4-heptanone (c) 2-Methylpentanal and...
-
Tell the prominent IR absorptions and mass spectral peaks you would expect for the following compound:
-
Explain possible advantages and disadvantages of PSL's current international trading strategy (subsidiary in China, export & import and online presence).
-
In November 2020 you entered into four May 2021 long futures contracts for crude oil (1,000 barrels per contract) for $65.00 per barrel. If the futures price was $70.00 per barrel on December 31,...
-
4) Champagne, Inc., had revenues of $13 million, cash operating expenses of $9 million, and depreciation and amortization of $2 million during 2008. The firm purchased $650,000 of equipment during...

Study smarter with the SolutionInn App


