What is the hybridization at each C in this molecule? Indicate the type of bond and the
Question:
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds?
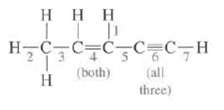
Transcribed Image Text:
Η Η Η ITT H=C=C=C=C_C7H (both) H tall three)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
1 OCsp2 H1s 5 OCsp2 Cs...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
What is the hybridization at the N and each C in this molecule? Indicate the type of bond and the orbitals that are overlapping to form it for each of the designated bonds (for example, ?CSP3 + H1s)....
-
Indicate the type of atomic orbitals that are overlapping to form each of the different kinds of bonds in CH3OCH3 (For example, a carbon sp3 AO and a hydrogen Is AO). What kinds of orbitals are...
-
Consider a tank containing a liquid, and the rate of change of the liquid's height (h) with respect to time (t) is proportional to the difference between the current height and a reference height....
-
Windsor General Hospital is a major hospital treating both private and public patients. It is a not for profit organization and its main sources of revenue are patient billing, state government...
-
Classify the following descriptions of constraints as bounds, limitations, requirements, proportional relationships, or balance constraints: a. Each serving of chili should contain a quarter pound of...
-
A round concrete storm sewer pipe used to carry rainfall runoff from a parking lot is designed to be half full when the rainfall rate is a steady \(1 \mathrm{in}\)./hr. Will this pipe be able to...
-
Bird-Bath, Inc., experienced four situations for its supplies. Compute the amounts that have been left blank for each situation. For situations 1 and 2, journalize the needed transaction. Consider...
-
Evaluate the integral. (Use C for the constant of integration.) 31dx 31 + ex Need Help? Read It Watch It Submit Answer
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Consider hydrogen cyanide, H C N. (a) What is the hybridization at the N at the C? (b) What are the types of the three CN bonds? What orbitals overlapping to form them? (c) In what type of orbital...
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it. : a) H_C_C,H 4 (both) : ...
-
This problem demonstrates the dependence of an annuitys future value on the size of the periodic payment. Suppose a fixed amount will be invested at the end of each year and that the invested funds...
-
Explain the role of interface in encapsulation. Provide examples.
-
Describe situations in which an organization should buy rather than build software components.
-
In interface design, what role do patterns play (or what role should they play)? Provide an example for patterns when you use an ATM to withdraw cash.
-
Explain the role of polymorphism in the user interface.
-
Identify objectives and solution features for the following products: a. An ATM. b. An accounting information system. c. A carwash. d. A car.
-
How many electrons are in the third shell of sodium, Na (atomic number 11)? (a) none (b) 1 (c) 2 (d) 3
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
Test the convergence of the series n=1 2n + 3 3 + 2
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
With regard to the production possibilities frontier (PPF), an efficient point is a point that is: ?
-
Within the context of rapidly evolving regulatory landscapes and heightened ethical scrutiny, how do advanced organizations navigate the ethical complexities of technological innovation and...
-
Write a program that accepts an integer limit and an integer value and that prints a complete line of output reporting the multiples of value uptil the limit . Keep prompting the user for a value...

Study smarter with the SolutionInn App


