Show the product, including stereochemistry, of the following reaction: 1. CH3MgBr, ether 2. H30*
Question:
Show the product, including stereochemistry, of the following reaction:
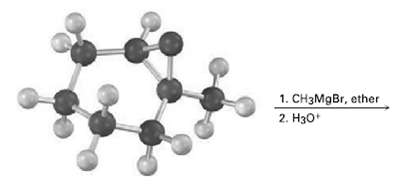
Transcribed Image Text:
1. CH3MgBr, ether 2. H30*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The Grignard reagent attacks the epoxid...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the product, including stereochemistry that would result from reaction of the following epoxide with HBr.
-
Show how a Wittig reaction can be used to prepare each of the following compounds. In each case, also show how the Wittig reagent would be prepared: (a) (b)
-
Product T is produced for $3.20 per pound, including a $0.20 per pound fixed cost. Product T can be sold without additional processing for $4.10 per pound, or processed further into Product U at an...
-
To load and register a driver, creating a new instance of the Driver class method is better than using the Class.forName() method. (True/False)
-
Describe why continuously learning about technology allows an executive to better analyze threats and opportunities.
-
A company that produces and markets continuing education programs on DVDs for the educational testing industry has traditionally mailed advertising to prospective customers. A market research study...
-
Give the formula and accompanying explanation of terms for computing achieved precision.
-
DJS Investment Services must develop an investment portfolio for a new client. As an initial investment strategy, the new client would like to restrict the portfolio to a mix of two stocks: The...
-
Stockman Corp, purchased ten $1,000 5% bonds of Simplex Corporation when the market rate of interest was 6%. Interest is paid semiannually, and the bonds will mature in nine years. Using the PV...
-
Ethical Dilemma Steven Sanchez is the export manager at Leon Industries, a manufacturer of office furnishings. He has identified Russia as a promising market and decides to attend a furniture trade...
-
Give IUPAC name for the following compounds (reddish brown =Br): (a) (b) (c)
-
Treatment of the following alkene with a peroxyacid yields an epoxide different from that obtained by reaction with aqueous Br2 followed by base treatment. Propose structures for the two epoxides,...
-
Why does merchandize inventory need to be adjusted at the end of the accounting periodand how is this done in a perpetual inventory system?
-
If a firm has a profit function where p=-120+ 200q-5q what output should it produce to maximize profit? What are profits for that q?
-
What is the breakeven output when the price is $6.40? To answer this question, a precise number is not required, but an interval is required (e.g., "the breakeven output is between X thousand and Y...
-
A recent graduate, 25 years old, is starting a career with an engineering firm. She intends to deposit a portion of her annual salary into an investment plan that returns 8% annually. Of the two...
-
What characteristics of Australia set its biodiversity apart from other global regions? Explain
-
What is an example of a specific perceptual error that you have experienced? This could be a time that someone misperceived something about you or someone around you or a time when you misperceived...
-
The following information relates to Karen Weigel Co. for the year 2025. Instructions After analyzing the data, prepare (a) an income statement and (b) a comprehensive income statement for the year...
-
Outline some of the major problems confronting an international advertiser.
-
In normal operation, a paper mill generates excess steam at 20 bar and 400C. It is planned to use this steam as the feed to a turbine to generate electricity for the mill. There are 5000 kg/hr of...
-
Show equations for the major fragmentations you would expect from the molecular ions of these compounds. List the m/z of the productions. CH3 CH2 a) CH;CH-CH,CH-CH; b) CH;CH,CHCH2CH3
-
(a) The base ion in the mass spectrum of 3-ethyl-2-methylpentane occurs at m/z 43m; show the fragmentation that produces this ion. (b) What other fragment would you predict to provide a major peak in...
-
Show equations to account for the major fragment ions that occur at the indicated m/z for thesecompounds: CH,CI b) CH3CH2CH,CH2OH nal: 31 miz 91 c) CH;CH,CCH,CH,CH,CH3 mtz 85, 72, 57
-
Image transcription text Introduction: The Smart Parking Management System (SPMS) project aimed to develop an efficient, automated solution for managing parking spaces in urban areas using Arduino...
-
A. Can we use human capital theory to explain "Age-Discrimination"? If so, how? B. Why do some firms prefer to hire younger workers, especially for entry level positions? C. Why do some firms...
-
Image transcription text Question 6 Not yet answered Marked out of 1.00 '1" Flag question During a mountain?biking tripr Bill and Barry are discussing the merits of having the springs and shocks used...

Study smarter with the SolutionInn App


