Show the products of thesereactions: CH,CI 1) NANH2, NH3 (/), NACN b) CH,C3C- a) 2) CH,CH,CH,Br DMSO
Question:
Show the products of thesereactions:
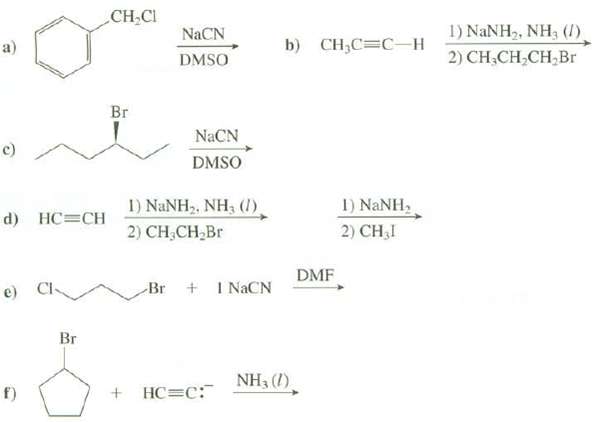
Transcribed Image Text:
CH,CI 1) NANH2, NH3 (/), NACN b) CH,C3C-н a) 2) CH,CH,CH,Br DMSO Br NaCN c) DMSO 1) NANH, 2) CH,I I) NANH2, NH, (1) d) HC=CH 2) CH,CH,Br DMF Br + I NACN e) Cl- Br NH3 (1). + HC=C: f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a CHCN e CL b ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
A financial institution can borrow $100 million for 2 years at 3%. It plans to invest this money in a 1-year security with an interest rate of 4.8% per year. Calculate net interest income for the...
-
Motomation Pty Ltd plans to acquire several retail automotive parts stores as part of its expansion program. Motomation carries out en extensive review of possible acquisitions prior to making any...
-
Ann Carter, Chief Financial Officer of Consolidated Electric Company (Con El), must make a recommendation to Con Els board of directors regarding the firms dividend policy. Con El owns two...
-
Discuss the seven stages of VM reviews.
-
Capri Wigs Company supplies wigs and hair care products to beauty salons throughout California and the Pacific Northwest. The accounts receivable clerk for Capri Wigs prepared the following partially...
-
Pina Colada Corporation manufactures car stereos. It is a division of Berna Motors, which manufactures vehicles. Pina Colada sells car stereos to Berna, as well as to other vehicle manufacturers and...
-
Describe the various types of transmission media.
-
Show the products of thesereactions: CH Br NaBH4 LIAIH4 ether b) a) CH,OH CH3
-
Suggest methods for preparing these compounds from alkylhalides: CN CH3 c) CH;C=CCH Ph b) HC=CCH,CH,CHCH3 a) "CH3
-
Assume you have just been hired as a business manager of PizzaPalace, a regional pizza restaurant chain. The companys EBIT was $120 million last year and is not expected to grow. PizzaPalace is in...
-
Electrostatic forces play an important role in biological systems, as many macromolecules, like DNA, are heavily charged ( see Example 1 3 ) . In fact, the electrostatic attractive force holds the...
-
In the following graphs, there is 3 graphs in a set. One graph is filled out for you. Fill out the shape of the other 2 graphs based on the given graph.
-
Draw a compartment model for a single organ where the rate into the organ is a constant and material is removed from the organ by radioactive decay ( where \ lambda represents the radioactive decay...
-
A solid sphere of diameter A is dropped in vacuum from a variable height H . At the bottom, you have a photosensor that measures the time interval ( DT ) between the bottom and top of the sphere...
-
A , B and C have masses of 2 9 9 . 5 , 5 3 4 . 6 , and 3 8 9 . 2 kg respectively. These masses are at the points ( 1 . 6 , 1 ) , ( 7 . 1 , 1 ) and ( 7 . 7 , 4 ) , where all distances are measured in...
-
Air at $2.5 \mathrm{kPa}, 221 \mathrm{~K}$ approaches the intake of a ramjet engine operating at an altitude of $25 \mathrm{~km}$. The Mach number is 3.0. For this Mach number, a normal shock stands...
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Find the slope of the line through each pair of points. (-12,-6), (0, -6)
-
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2: NaN3 (a) CHCH2CH2H2Br CH3CH2CH2CH,N=N=N THE CI (b) , CCH-CHCH2CH CHCH2CHCHCH3 Ethanol (c) H CI -CH -- -CH (d)...
-
Write the product you would expect from reaction of each of the following alkyl halides with (i) Na + ? SCH3 and (ii) Na + ?? OH (yellow-green = Cl): (a) (c) (b)
-
From what alkyl bromide was the following alkyl acetate made by SN2 reaction? Write the reaction, showing allstereochemistry.
-
The blue samurai, a japanese restraurant, has an asset turnover of 3.5 the total assets were 95,000 what are net sales for the blue samurai?
-
Gatekeeper Manufacturing reported 50,000 physical units that were 100% complete for direct materials during the period. In addition, the 50,000 physical units were 100% for conversion costs. In terms...
-
What red flag was overlooked on the Montague Fellowship Expense Report?

Study smarter with the SolutionInn App


