Show the products of thesereactions: CH3 . b) . CH,N2 CH-l2 a) C=C C=C Zn(Cu)
Question:
Show the products of thesereactions:
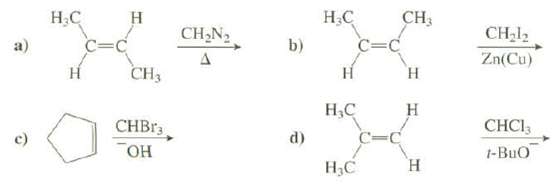
Transcribed Image Text:
CH3 Н.С b) Н.С н CH,N2 CH-l2 a) C=C C=C Zn(Cu) Н Н Н CH3 Н.С н CHBR3 CHCI3 t-BuO d) Он Н,С н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
CH a HCCC H ...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
1. Which type contains a single character enclosed within single quotes? A. Character B. Numeric C. Floating point 2. The modulus operator uses, B. - B. < A. + 3. Every variable should be separated...
-
1. How can location-based analytics help retailers in targeting customers? 2. Research similar applications of location-based analytics in the retail domain.
-
How much would be appropriate for wealthy countries to contribute to global health? Why should we do more than we do now? What's in it for us?
-
What is the Developmental Configuration?
-
Golden Gardens Inc. develops and produces spraying equipment for lawn maintenance and industrial uses. On June 19 of the current year, Golden Gardens Inc. reacquired 24,000 shares of its common stock...
-
Mauve Company uses a standard cost system that applies manufacturing overhead to units of product on the basis of direct labour hours (DLHS). The following data pertain to last month: Actual Hours...
-
The financial statements of Valerie's Natural Foods include the following items: Compute the following ratios for the current year: a. Current ratio b. Cash ratio c. Acid-test ratio d. Inventory...
-
Explain how a similar hydroboration reaction could be used to prepare (R)-2-butanol in good enantiomeric excess.
-
Show the products of thesereactions: CH3 . PHCO;H MCPBA NaOH b) C= a) ,
-
Sketch the graph of the equation by point plotting. y = 1/2x + 2
-
How do mindfulness-based interventions, rooted in principles of attention regulation and non-reactivity, offer novel avenues for cultivating resilience and fostering adaptive stress responses across...
-
A house is sold for $195,000. The mortgage is $168,745.60 at 8%. Annual taxes are $3,893.25. The closing will occur on June 15. What are the total prorations for interest (for the full month) and for...
-
Can you delve into the neurobiological underpinnings of stress and the implications for developing targeted interventions aimed at mitigating its adverse effects on physiological and psychological...
-
Consider the frame shown below and answer the questions that follow. 3 a 12 2 4 3 1 b 0 P = 4 2 0 2 5 0 0 0 1 Find the missing values, a and b. Explain your reasoning.
-
Complete the following exercises by applying polynomial identities to complex numbers. Show your work. Factor x 2 + 64. Check your work. Factor 16x 2 + 49. Check your work. Find the product of (x +...
-
A thermal reservoir has entropy gradient \(d S / d E=b E^{3}\), where \(b=2.00 \mathrm{~J}^{-4}\). At \(E=3.00 \mathrm{MJ}\), what is the temperature of the reservoir?
-
Evenflow Power Co. is considering a new project that is a little riskier than the current operations of the company. Thus, management has decided to add an additional 1.5% to the company's overall...
-
A 0.100-g sample of the weak acid HA (molar mass = 100.0 g/mol) is dissolved in 500.0 g water. The freezing point of the resulting solution is -0.0056C. Calculate the value of K a for this acid....
-
Propose a mechanism for the followingreaction: HO- CO2CH3 (CH3CH2)3N Heat BRCH2 CO2CH3
-
One step in the biosynthesis of morphine is the reaction of dopamine with p-hydroxyphenylacetaldehyde to give S)-norcoclaurine. Assuming that the reaction is acid-catalyzed, propose amechanism. . NH ...
-
The antitumor antibiotic mitomycin C functions by forming cross-links in DNA chains. (a) The first step is loss of methoxide and formation of an iminium ion intermediate that is deprotonated to give...
-
We review long-lived assets for impairment when circumstances indicate the carrying amount of an asset may not be recoverable based on the undiscounted future cash flows. If the carrying amount of...
-
Hal's Heavenly Creations offers its employees the option of contributing up to 6% of their salaries to a voluntary retirement plan, with the employer matching their contribution. The company also...
-
Altira Corporation provides the following information related to its inventory during the month of August 2024: August 1 Inventory on hand-2,300 units; cost $6.40 each. August 8 Purchased 11,500...

Study smarter with the SolutionInn App


