Show the products of thesereactions: Br CH,OH CH3CH,CHCH,CH, + CH,0 CH,CH-CHCH,CH3 Br CH;OH CH,CHCH,CH,CH; + CH,0 CH.CH-CHCH,CH;
Question:
Show the products of thesereactions:
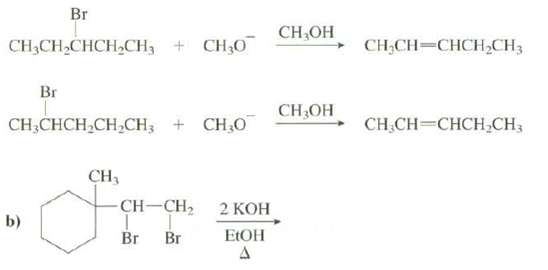
Transcribed Image Text:
Br CH,OH CH3CH,CHCH,CH, + CH,0 CH,CH-CHCH,CH3 Br CH;OH CH,CHCH,CH,CH; + CH,0 CH.CH-CHCH,CH; CH, CH-CH2 2 КОН b) ELOH Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a C...View the full answer

Answered By

Shivani Dubey
Hello guys, I am very good with the Advance mathematics, Calculus, Number theory, Algebra, Linear algebra, statistics and almost all topics of mathematics. I used to give individual tutoring students in offline mode but now want to help students worldwide so I am joining here. I can also give solutions in various coding languages of mathematics and software like mathematica, R, Latex, Matlab, Statistica, etc.
feel free to ask any doubt regarding mathematics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
What is the significance of communication between managers and their subordinates, and how does it impact employee performance?
-
Colonial Tap Company (CTC) is a manufacturer of taps and fittings for the plumbing trade, located in Brisbane. The company was established by Ken Hall in 1951, with a workforce of 10, to meet the...
-
You receive a brochure from a large university. The brochure indicates that the mean class size for full-time faculty is fewer than 32 students. You want to test this claim. You randomly select 18...
-
Define value management (VM) and its key principles and attributes.
-
Refer to Problem 12-1. What would be the additional funds needed if the companys year-end 2010 assets had been $4 million? Assume that all other numbers, including sales, are the same as in Problem...
-
2. Data Types: Your Moose Jaw Reservoir Team in Saskatchewan has collect well cores, well logs and seismic data (acoustic impedance). Share your knowledge with your team. (3 points) a) What is the...
-
Every college student had the problem of selecting the college or university to attend. Was this a simple, intermediate, or complex problem for you? Explain.
-
Explain which of these reactions would provide a better synthesis of2-pentene: Br CH,OH CH,CH,CHCH,CH, + CH;0 CH,CH=CHCH,CH3 Br CH, CH CH=CHCH CH, CH,CHCH,CH,CH; + CH,0
-
Show the products of thesereactions: CH, OH CH3 H,SO, H,SO, b) H3CCOH H PO, CH,CH-CHCH, H;SO4 a) CH
-
a. Information gathering: Prepare a list of questions that you will inquire of management at the afternoon meeting regarding this new development. b. Analysis and evaluation: Assuming this new...
-
1. *For following circuit where C = C = 100 uF, R = R = 2000. ww R x(1) R a) Find its transfer function H(s). b) Find its poles. c) Find its unit impulse response h(t). C y(t)
-
Assume a Centaur V upper stage with an empty mass of 2,247 kg, diameter of 3.05 meters and height of 12.68m, the wall thickness is 41mm, whereas the motor can be assumed to fill up 1/4 of the inner...
-
For pressure-driven laminar flow between two horizontal infinite parallel plates separated by a distance 2h, the velocity components are: a) where U is the centreline velocity and the x-axis is...
-
Discuss how geometry, surface condition and material quality can have; a) A significant influence on the fatigue life of a cyclically stressed component. (5 marks) b) Explain the differences between...
-
Laura dives with a watch around her arm. Her watch can withstand an absolute pressure of P = 5.5 bar? Assume a gravity acceleration of g = 9.81 m/s, and a density of water pw = 1002 kg/m. Hint: 1 bar...
-
Air enters a combustion chamber at $75 \mathrm{~m} / \mathrm{s}, 150 \mathrm{kPa}$ and $300 \mathrm{~K}$. Heat addition in the combustor amounts to $900 \mathrm{~kJ} / \mathrm{kg}$. Compute (a) the...
-
Data 9.2 on page 540 introduces the dataset Cereal, which includes information on the number of grams of fiber in a serving for 30 different breakfast cereals. The cereals come from three different...
-
What is probability of getting two heads in a row flipping a coin three times?
-
Ethers can often be prepared by S N 2 reaction of alkoxide ions, RO ? , with alkyl halides. Suppose you wanted to prepare cyclohexyl methyl ether. Which of the two possible routes shown below would...
-
We saw in Section 7.8 that bromohydrin are converted into epoxides when treated with base. Propose a mechanism, using curved arrows to show the electronflow. Br -C CH3 CH NAOH Ethanol TH CH C
-
Show the stereochemistry of the epoxide you would obtain by formation of a bromohydrin from trans-2-butene, followed by treatment with base.
-
Melissa Cutt is thinking about buying some shares of EZLawn Equipment, at $36.44 per share. She expects the price of the stock to rise to $43.62 over the next 3 years. During that time she also...
-
At the beginning of the week, Taylan's restaurant had a beginning inventory of food and beverages totaling $4,000. During the week, additional food and beverages purchases totaled $3,000. At the end...
-
1. Identify the auditor's role in the internal and external auditing processes. 2. Identify one law, regulation, and statue affecting healthcare

Study smarter with the SolutionInn App


