Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO.
Question:
Show the structures of A, B, C, and D in the following reactionsscheme:
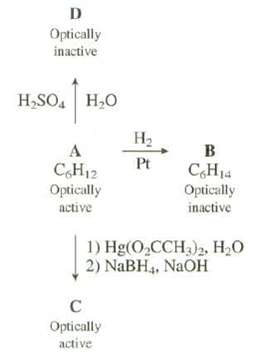
Transcribed Image Text:
D Optically inactive H,SO. H,O На B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH Optically active
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
OH optically inactive D ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the structures of the carbocation intermediates you would expect in the following reactions: (b) CH3 (a) CH3CH2CCHCH3 HI CH H Er
-
Show the structures of the products you would obtain by hydroboration/oxidation of the following alkenes: H (a) "CH (a) CH3C=CHCH2CH3 (b)
-
Show the structures of the likely fragments you would expect in the mass spectra of the followingmolecules: (a) (b)
-
Benny sells an apartment building. His adjusted basis for regular income tax purposes is $450,000, and it is $475,000 for AMT purposes. He receives $700,000 from the sale. a. Calculate Bennys gain...
-
Gresham College is a local private college. When reviewing the colleges financial reports you would expect to see which of the following categories on its statement of assets and liabilities?
-
Closing slows down the recording of next years transactions. Defend or reject this statement with supporting evidence.
-
What are the two classes of OE domains?
-
On December 31, after adjustments, Gomez Companys ledger contains the following account balances: 101 Cash .................$47,200 Dr. 111 Accounts Receivable .......... 17,800 Dr. 121 Supplies...
-
Marlow Company uses a perpetual inventory system. During the year, it entered into the following purchases and sales transactions. Date Activities Jan. 1 Beginning inventory Feb. 10 Purchase Mar. 13...
-
At high temperatures, alkanes can undergo dehydrogenation to produce alkenes. For example: This reaction is used industrially to prepare ethylene while simultaneously serving as a source of hydrogen...
-
Limonene, a major component of lemon oil, has the formula C10H16. (a) On reaction with excess H2 in the presence of Pt, limonene produces C10H20. What information does this provide about the...
-
In Figure, suppose Br2 adds to the alkene from the bottom, rather than from the top as shown. Analyze the stereochemistry of the reaction in this case and explain which products areformed. Relative...
-
Some financial data for three corporations are displayed here. a. Which firm appears to be excessively leveraged? b. Which firm appears to be employing financial leverage to the most appropriate...
-
Noble issued preferred stock with a $9 dividend per year. The investment bank paid Noble $72 per share for the stock and then sold the stock to investors for $75 per share. a. If you buy the stock...
-
After seeing a presentation on the power of data mining and hearing about your past consulting work, you are approached to create a model for a university's admissions office. The members of the...
-
Recently you borrowed money for a new car. The loan amount is $15,000 to be paid back in equal annual payments which begin today, and will continue to be payable at the beginning of each year for a...
-
What is the monthly repayment on a $300,000 home loan with a term of 25 years at a fixed interest rate of 6% pa?
-
Assume today is an earnings announcement day for a firm. For the day, the firm's return was .8 percent, while the risk-free daily rate was .01 percent and the market rate of return was 1.1 percent....
-
An automobile manufacturer determines that, on a summer day when the ambient temperature is \(311 \mathrm{~K}\), the temperature inside their new, ready-to-deliver cars reaches \(339 \mathrm{~K}\)...
-
Suppose that the electrical potential at the point (x, y, z) is E(x, y, z) = x + y - 2z. What is the direction of the acceleration at the point (1,3,2)?
-
Sulfuryl chloride undergoes first-order decomposition at 320.C with a half-life of 8.75 h. What is the value of the rate constant, k, in s -1 ? If the initial pressure of SO 2 Cl 2 is 791 torr and...
-
Give the amino acid sequence of hexapeptides that produce the following sets of fragments on partial acid hydrolysis: (a) Arg, Gly, IIe, Leu, Pro, Val gives Pro-Leu-Gly, Arg-Pro, Gly-IIe-Val (b) N,...
-
Show the mechanism for formation of a Boc derivative by reaction of an amine acid with di-tert-butyl dicarbonate.
-
Write all five steps required for the synthesis of Leu-Ala from alanine and leucine.
-
Doing a strategic analysis of GraceKennedy Limited, What is the current level of its economic performance, an indication of the factors responsible for the current performance and recommendations for...
-
How would you evaluate Cisco social media marketing success? Outline which, if any, of the mentioned media plans Cisco should pursue. Why? What are the challenges of using social media? Do you have...
-
Office Ltd manufactures furniture for computer work stations. The company uses a job costing system. Manufacturing overhead is applied to production at a predetermined overhead rate of $60 per direct...
Discrete Random Signals And Statistical Signal Processing 1st Edition - ISBN: 0138521123 - Free Book

Study smarter with the SolutionInn App


