Show the structures of the carbocation intermediates you would expect in the following reactions: (b) CH3 (a)
Question:
Show the structures of the carbocation intermediates you would expect in the following reactions:
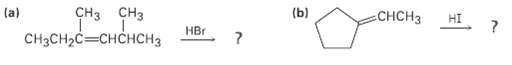
Transcribed Image Text:
(b) СНCH3 (a) CH3CH2C—CHCHСН3 HI CHз сHз НEr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The more stable carbocation is formed a ...View the full answer

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the structures of the likely fragments you would expect in the mass spectra of the followingmolecules: (a) (b)
-
Compare the structures of a carbocation and a carbanion: In one of these ions, the central carbon atom is trigonal planar; in the other it is trigonal pyramidal. Assign the correct geometry to each...
-
Show the structures of the products you would obtain by hydroboration/oxidation of the following alkenes: H (a) "CH (a) CH3C=CHCH2CH3 (b)
-
A particle is described by the normalized wave function (x, y, z) = Axe-ax2 e -y2 e-yz2', where A, a, , and are all real, positive constants. The probability that the particle will be found in the...
-
1. Based on the limited evidence, how would you characterize Frank's leadership style? 2. Which leadership behaviors and attitudes is Frank displaying? 3. How else might Frank approach his project...
-
Show how the following floating-point additions are performed (where significands are truncated to 4 decimal digits). Show the results in normalized form. a. 5.566 102 + 7.777 102 b. 3.344 101 +...
-
What is a beat frequency?
-
The State of Ohio decides that Ohio's landfills are becoming too full at too rapid a pace, so it passes a law banning the import of waste generated out of state. Several landfill operators have...
-
Two sound sources generate pure tones of 81 Hz and 56 Hz. What is the beat frequency?
-
A bicycle wheel of radius R and mass M is at rest against a step of height 3R/4, as illustrated in FIGURE 11-65. Find the minimum horizontal force F that must be applied to the axle to make the wheel...
-
What alkenes would you start with to prepare the following alkylhalides? CH-CH (a) (b) Br Br CI (c) (d) CH3CH2CHCH2CH2CH3
-
Draw a skeletal structure of the following carbocation. Identify it as primary, secondary, or tertiary, and identify the hydrogen atoms that have the proper orientation for hyper conjugation in the...
-
Simplify the radical expressions in Exercises 6774 if possible. 162x5 2x
-
Write a memo addressed to Misters Conner and Martin, evaluating the risk of CMC and discussing the pros and cons of debt versus equity financing. Base your analyses on financial ratios calculated...
-
A marketing database contains: Group of answer choices transactions and interactions individuals have with a firm. information about current customers and customers of the competition. customer...
-
Identify a few of the internal and external governance mechanisms in operation today in business. Why do good people sometimes act in an unethical manner even with these control mechanisms in place?
-
Explain the concept of statutory job protections. Are casual employees subject to such protections
-
In 2022, Keith rode in a vanpool to work. Keith's employer paid Keith $5,000 for Keith's participation in the vanpool. The vanpool was a qualified transportation fringe benefit provided by Keith's...
-
J. Hill has the following assets and liabilities on 30 November 2016: Accounts payable 2,800; Equipment 6,200; Car 7,300; Inventory 8,100; Accounts receivable 4,050; Cash at bank 9,100; Cash in hand...
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
Benzene has a heat of vaporization of 30.72 kJ/mol and a normal boiling point of 80.1 C. At what temperature does benzene boil when the external pressure is 445 torr?
-
Using the curved-arrow notation, derive a resonance structure for the allyl anion (shown here) which shows that the two carbon-carbon bonds an identical bond order of 1.5 and that the unshared...
-
Using the curved-arrow notation, derive a resonance structure for the allyl anion (shown here) which shows that the two carbon-carbon bonds an identical bond order of 1.5 and that the unshared...
-
Using Table 3.1, as well as the data given below, estimate the equilibrium constants for the following reactions at 25C. pK, 10.5
-
Annuity Payments Go to www.fcfcorp.com/onlinecalc.htm. Use the calculator to solve this problem. If you have $1,500,000 when you retire and want to withdraw an equal amount for the next 30 years, how...
-
Calculating Future Values Go to www.dinkytown.net and follow the Savings Calculator link. If you currently have $10,000 and invest this money at 9 percent, how much will you have in 30 years? Assume...
-
In 2023, Amanda and Jaxon Stuart have a daughter who is 1 year old. The Stuarts are full-time students and are both 23 years old. Their only sources of income are gains from stock they held for three...

Study smarter with the SolutionInn App


