Limonene, a major component of lemon oil, has the formula C10H16. (a) On reaction with excess H2
Question:
Limonene, a major component of lemon oil, has the formula C10H16.
(a) On reaction with excess H2 in the presence of Pt, limonene produces C10H20. What information does this provide about the structure of limonene?
(b) On ozonolysis, limonene produces these compounds. Suggest possible structures forlimonene.
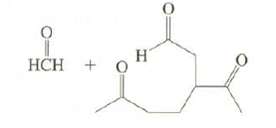
Transcribed Image Text:
Н НСН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a The DU of limonene is 3 The DU of the hydrogena...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A major component of financial planning is to forecast future financial statements. If you had a company's balance sheets and income statements for the past 5 years but no other information, how...
-
A major component of gasoline is octane (c8h18). When liquid octane is burned in air it reacts with oxygen (o2) gas to produce carbon dioxide gas and water vapor. Calculate the moles of oxygen needed...
-
1- Explain why all the amino acids except for glycine are chiral? 2-Identify the amino acids that differ from each other by a single methyl or methylene group.? 3-Classify the 20 standard amino acids...
-
According to the American Red Cross, 11.6% of all Connecticut residents have Type B blood. A random sample of 28 Connecticut residents is taken. X = the number of Connecticut residents that have Type...
-
Grania knows that the pH of blood is normally 7.35 to 7.45. She sees that her blood test results show 7.57. Is Granias blood too acid or too alkaline or normal?
-
What role can internal marketing play in enhancing effectiveness of both planning and implementation?
-
Discuss what corporate governance is and why it is important to shareholders.
-
On January 2, 2015, Ditto Clothing Consignments purchased showroom fixtures for $12,000 cash, expecting the fixtures to remain in service for depreciated the fixtures on a double-declining-balance...
-
Compare the static budget variances as provided in Exhibit 3 and the flexible budget variances you computed in Questions 5 to 8. Assume that the production manager is primarily responsible for...
-
1. What are the main reasons for the success of ZamZam Cola in Middle East? 2. How should Ahmad-Haddad Moghaddam prepare his sales force culturally for selling ZamZam Cola to European supermarket...
-
Suggest a mechanism for thisreaction: CH,Br Br2 CH2=CHCH CH,CH,OH H,O
-
Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO. H,O B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH...
-
Rosendale Clothiers has decided to make significant reinvestments in its operations. As a result it will be suspending dividend payments for 2 years. It anticipates paying a dividend again in year 3...
-
Convert the following 32-bit IEEE bit patterns to their corresponding floatingpoint values: (a) 0 10000000 10010010000111111011011 (b) 0 10000000 01011011111100001010100 (c) 0 01111111...
-
A haulage firm uses containers to carry products for clients which currently measure 10 m long by 2 m wide by 4 m high. The owner of the firm has decided to invest in new containers which double...
-
Write a function to count the number of 1 bits in an unsigned 32-bit integer.
-
This chapter discusses companies that are oligopolists in the market for the goods they sell. Many of the same ideas apply to companies that are oligopolists in the market for the inputs they buy. If...
-
In an x-ray diffraction experiment on a crystal that has a cubic lattice, the smallest Bragg angle at which you see a peak in the intensity of diffraction pattern is \(15.00^{\circ}\). If the...
-
What are the three levels of international strategy? Why is it important to distinguish among the levels?
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
A popular chemical demonstration is the magic genie procedure, in which hydrogen peroxide decomposes to water and oxygen gas with the aid of a catalyst. The activation energy of this (uncatalyzed)...
-
The octapeptide angiotensin II has the sequence Asp-Arg-Val-Tyr-IIe-His-Pro-Phe. What fragments would result if angiotensin II were cleaved with trypsin with chymotrypsin?
-
What is the N-terminal residue on a peptide that gives the following PTH derivative on Edmandegradation?
-
Draw the structure of the PTH derivative that would he formed on Edman degradation of angiotensin II (Problem 26.12).
-
what is the meaning of each variable in capital asset pricing model equation?
-
Further to the lesson discussions and readings about the Sales and Operations Planning Process, provide your argument for the value of this process applied to a CPG (Consumer Packaged Goods) company....
-
You are a financial advisor to a client who just made a lot of money on an IPO recommended to him by a friend. Now your client wants to make dramatic changes to his portfolio, switching from a...

Study smarter with the SolutionInn App


