What alkenes would you start with to prepare the following alkylhalides? CH-CH (a) (b) Br Br CI
Question:
What alkenes would you start with to prepare the following alkylhalides?
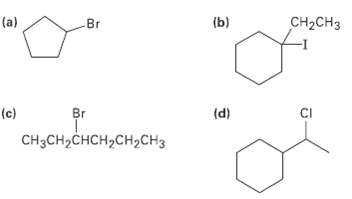
Transcribed Image Text:
CH-CHз (a) (b) Br Br CI (c) (d) CH3CH2CHCH2CH2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
Strategy Think backward in choosing the alkene starting material for synt...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkynes would you start with to prepare the followingketones? (a) (b) CHCHH2H CH3CH2CH2CH3
-
What alkyne would you start with to prepare each of the following compounds by a hydroboration/oxidationreaction? (b) (a) CH CH-CCHCH3 -CH2CH CH
-
What alkenes would you choose as starting materials in order to prepare the following cyclopropane derivatives by reaction with iodomethylzinc iodide?
-
A beam of 40-eV electrons traveling in the +x-direction passes through a slit that is parallel to the y-axis and 5.0wide. The diffraction pattern is recorded on a screen 2.5 m from the slit. (a) What...
-
1. What would you advise Tricia to do about future posts of a personal nature to the team? 2. Which one or two relation-oriented behaviors does Tricia appear to be exagerrating? 3. If your team...
-
The Zwerling family installed central air conditioning in their house this summer. They are comparing the electric bills of this summer and last summer. The data is shown. a. What was the total...
-
What is a system?
-
List the pressures that lean systems pose for supply chains, whether in the form of process failures due to inventory shortages or labor stoppages, etc. Reflect on how these pressures may apply to a...
-
1. How does the Bail system affect Minority? 2. How does the Bail System violate our constitutional rights? 3. How does the Bail System violate our 8 th and 14 th Amendments? 4. How is the Bail...
-
What type of processproject, job shop, flow shop, and continuous flowwould most likely be used to produce the following? Explain your reasoning. a. Apple iPads b. Weddings c. Paper d. Tax preparation
-
Predict the products of the following reactions: (b) CH CHCCHH2CH (a) HCI H (d) CH CH2 (c) HBr CH-CH3CH2 H20 H2SO, (Addition of H20 occurs.)
-
Show the structures of the carbocation intermediates you would expect in the following reactions: (b) CH3 (a) CH3CH2CCHCH3 HI CH H Er
-
In the hotel industry, package rate refers to a a. Group of hotel products and services sold for one price. b. Room rate discount offered to members of a consortium. c. Room that is sold at full, or...
-
For Kosko Company, actual sales are $1.218,000 and break-even sales are $962,220. Compute (a) the margin of safety in dollars and (b) the margin of safety ratio. Margin of safety Margin of safety...
-
4 Smallman's Garage uses standards to plan and control labor time and expense. The standard time for an engine tune-up is 3.75 hours. and the standard labor rate is $15 per hour. Last week, 26...
-
The following data were gathered to use in reconciling the bank account of Bradford Company: Balance per bank $ 18,050 Balance per company records 10,040 Bank service charges 50 Deposit in transit...
-
. Assume that over the past 88 years, U.S. Treasury bills had an average return of 3.5% as compared to 6.1% on long-term government bonds. What was the average risk premium on the long-term...
-
J is going to receive a 30-year annuity of 8,500 and L is going to receive perpetuity of 8,500. If the appropriate interest rate is 6%, how much more is L's cash flow worth?
-
In what way is the greenhouse effect like a florists greenhouse?
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Carbon disulfide has a vapor pressure of 363 torr at 25 C and a normal boiling point of 46.3 C. Find Hvap for carbon disulfide.
-
The energy difference between anti-butane and either one of the gauche-butane conformations is 2.8 kj mol-1(0.67 kcal mol-1). Treating this difference as a standard free energy, calculate the amounts...
-
The energy difference between anti-butane and either one of the gauche-butane conformations is 2.8 kj mol-1(0.67 kcal mol-1). Treating this difference as a standard free energy, calculate the amounts...
-
Arrange the compounds in each of the following sets in order of decreasing pKa, highest first. Explain your reasoning. (a) CLCH2CH2SH CH3CH2OH CH3CH2SH (b) CH,CH,OH (CH3),N-CH-CH,OH (CH3)N OH
-
On March 1, 2017 Green Company purchased a new piece of equipment for 210,000 cash. On March 1 Green recorded the equipment purchase with a Debit to the Equipment account and a Credit to the Cash...
-
The price per share of your all-equity firm is $40, and there are 2M shares outstanding. Suppose that your firm issues $30M worth of debt. The debt has a face value of $30M, a coupon rate of 5...
-
Provide a detailed explanation explaining the importance of organizational politics in the workplace. I am providing two references that you can use in your response. Please provide the pros and cons...

Study smarter with the SolutionInn App


