Use the data in Appendix 2 to predict whether or not the following reactions are feasible. If
Question:
Use the data in Appendix 2 to predict whether or not the following reactions are feasible. If a reaction does occur, write a balanced equation for it.
a. Can MnO4 – ions oxidise Cl– ions to Cl2 in acidic conditions?
b. Can MnO4– ions oxidise F– ions to F2 in acidic conditions?
c. Can H+ ions oxidise V2+ ions to V3+ ions?
d. Can H+ ions oxidise Fe2+ ions to Fe3+ ions?
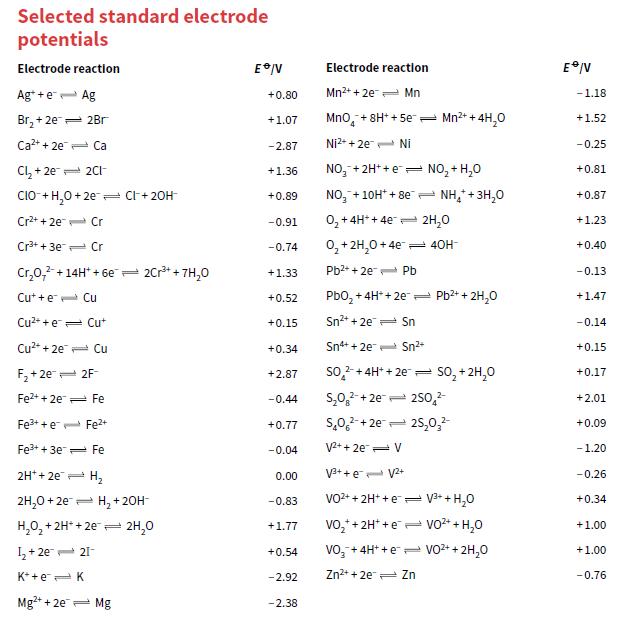
Transcribed Image Text:
Selected standard electrode potentials Electrode reaction E/V Electrode reactlon Ag* +e- Ag Mn2+ + 2e= Mn +0.80 -1.18 Br, + 2e 2Br Mno, + 8H* + 5e Mn2+ + 4H,0 +1.52 +1.07 Ca2+ + 2e Ca -2.87 Ni24 + 2e NI -0.25 Cl, + 2e - 201 NO, + 2H* + e - NO, + H,0 +1.36 +0.81 CIO + H,0 + 2e"= - Cl+ 20H- NO, + 10H* + 8e" NH,* +3H,0 +0.89 +0.87 Cr2+ + 2e Cr -0.91 0,+4H* + 4e 2H,0 +1.23 Cr3+ + 3e Cr 0,+ 2H,0+ 4e= 40H- -0.74 +0.40 Cr,0,7 +14H* + 6e 2Cr* + 7H,0 Pb2+ + 2e Pb -0.13 +1.33 Cut +e Cu Pbo, + 4H* + 2e - Pb2+ + +1.47 +0.52 Cu2+ +e= Cut +0.15 Snt + 2e Sn -0.14 Cu2+ + 2e Cu Sn+ + 2e Sn2+ +0.34 +0.15 F,+ 2e"- 2F- so + 4H* + 2e = so, + 2H,0 +0.17 +2.87 Fe2+ + 2e Fe -0.44 s,0,2 +2e- 25o, +2.01 Fe+ +e Fe2+ 5,0,2- + 2e= 25,0, +0.77 +0.09 Fe+ + 3e Fe -0.04 V2+ + 2e= V -1.20 2H* + 2e H, V3+ +e- V2+ 0.00 -0.26 2H,0 +2e VO2+ + 2H* +e V3+ + H,0 H, +20H- -0.83 +0.34 H,0, + 2H* + 2e - 2H,0 vo,* + 2H* +e= vo2* + H,0 +1.77 +1.00 I + 2e 21- vo,, + 4H* +e= vo2+ + 2H,0 +0.54 +1.00 K* +e= K -2.92 Zn2+ + 2e Zn -0.76 Mg2+ + 2e= Mg -2.38
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
To predict whether a reaction is feasible or not we need to compare the standard reduction potentials of the two halfreactions involved in the overall reaction If the reduction potential ...View the full answer

Answered By

Shazni Ahamed
my self sudhanshu pandey , i have two year teaching experience . while teaching i have learned how to teach intresting way so that student can understand easily and they can retain for long time.And i can easily understand student problem
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Predict whether or not He2+ exists.
-
Write the balanced equation for the sequential conversion of glucose to pyruvate and of pyruvate to glucose.
-
Use the data in Table 19.1 to determine whether or not hydrogen peroxide will undergo disproportionation in an acid medium: 2H2O2 2H2O + O2.
-
Let n=102 +r, where , re N, 0r9. A number a is chosen at random from the set {1, 2, 3, ..., n} and let pn denote the probability that (a 1) is divisible by 10. 70. If r = 0, then npn equals (a) 22...
-
Let z be a random variable with a standard normal distribution. Find the indicated probability and shade the corresponding area under the standard normal curve. P(0.45 z 0.54)
-
You work for Nokia in its global cell phone group. You have been made project manager for the design of a new cell phone. Your supervisors have already scoped the project so you have a list showing...
-
If Exercise 8.3 had asked about a book in free fall rather than one on the floor, what would the free-body diagram look like? Data from Exercise 8.3 Draw a free-body diagram for a book lying...
-
The capital investment committee of Ellis Transport and Storage Inc. is considering two investment projects. The estimated income from operations and net cash flows from each investment are as...
-
4. An ideal gas undergoes a thermodynamic process in which internal energy aP4, where a is a (U) of the gas depends on pressure (P) of the gas as =
-
In this lab, we will use the inverse kinematics to identify two functions to generate an animation of a two-link robot. As mentioned in the previous section, the lengths of the two links are a = 3 ft...
-
State the direction of the electron flow in the electrochemical cells represented by the following pairs of half-equations. Use the data in Appendix 2 to help you. a. F 2 + 2e 2F and Mn 2+ + 2e ...
-
Suggest a suitable reagent that can carry out each of the following oxidations or reductions. Use the data in Appendix 2 to help you. a. The reduction of Zn 2+ ions to Zn. b. The oxidation of Br ...
-
An electric heater is used to heat a room of volume 62m3. Air is brought into the room at 5oC and is completely replaced twice per hour. Heat loss through the walls amounts to approximately...
-
Nebraska Inc. issues 4,750 shares of common stock for $152,000. The stock has a stated value of $14 per share. The entry to journalize the stock issuance would include a credit to Common Stock for
-
If an employee option to buy 4,000 shares of the employer's stock has a strike price of $15.50 and on May 29, 2021 the stock's market price is $29.90, what is the intrinsic value of this option?...
-
A customer is angry. He/She does not like a service provided by a company. He/She claims his/her money back. He/she want to get 100% of this sum. According to a contract a customer signed with a...
-
When the data entry is done, the program will display a list of options as follow. 1 Search test result by applicant Id 2 Search test result(s) by applicant name 3 Search test result(s) by overall...
-
From The American Scholar we have the following quote: In the degenerate state, when the victim of society, he [the scholar] tends to become a mere thinker, or, still worse, the parrot of other men's...
-
Compare the orbital radii of the He+ and H atoms for levels of equal energy (not the same value of n). Can you draw a general conclusion from your results?
-
Write a function that reads a Float24_t value: Float24_t float24_read(void) A legitimate float24 value string is of the form: "mantissabexponent" where the mantissa (m) and the exponent (e) may have...
-
Show how you would use an alkoxymercuration-demercuration to prepare isopropyl propyl ether using propene as your only source of carbon and any other reagents of your choosing.
-
When Julius Caesar expired, his last exhalation had a volume of 450. cm 3 and contained 1.00 mole percent argon. Assume that T = 300.K and P = 1.00 atm at the location of his demise. Assume further...
-
Predict the products for each of the following reactions: a. b. c. d. e. f. excess HBr heat excess HI heat
-
Product/service features and benefits for athlete pharmacy-medical supply Describe all of your products or services, being sure to focus on the customer's point of view. For each product or service:...
-
As a result of the therapeutic misconception, subjects often "deny the possibility that there may be major disadvantages to participating in clinical research that stem from the nature of the...
-
6. For this K-Map: ab 00 01 11 10 00 0 0 0 0 PD 01 0 0 1 0 cd 11 1 0 0 1 10 0 1 0 0 a. What are the implicants? (2 points) b. What are the prime implicants? (2 points) c. What are the essential prime...

Study smarter with the SolutionInn App


