A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in
Question:
A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming there is no heat transfer between the gas and the tank, find the pressure in the tank at the end of the venting process and the fraction of the initial mass of gas remaining in the tank for each of the following cases.
a. The equation of state of carbon dioxide is P(V − b) = RT with b = 0.0441 m3/kmol
b. Carbon dioxide obeys the law of corresponding states of Sec. 6.6.
c. Carbon dioxide obeys the Peng-Robinson equation of state. The low-pressure (ideal gas) heat capacity of CO2 is given in Appendix A.II.
Appendix A.II
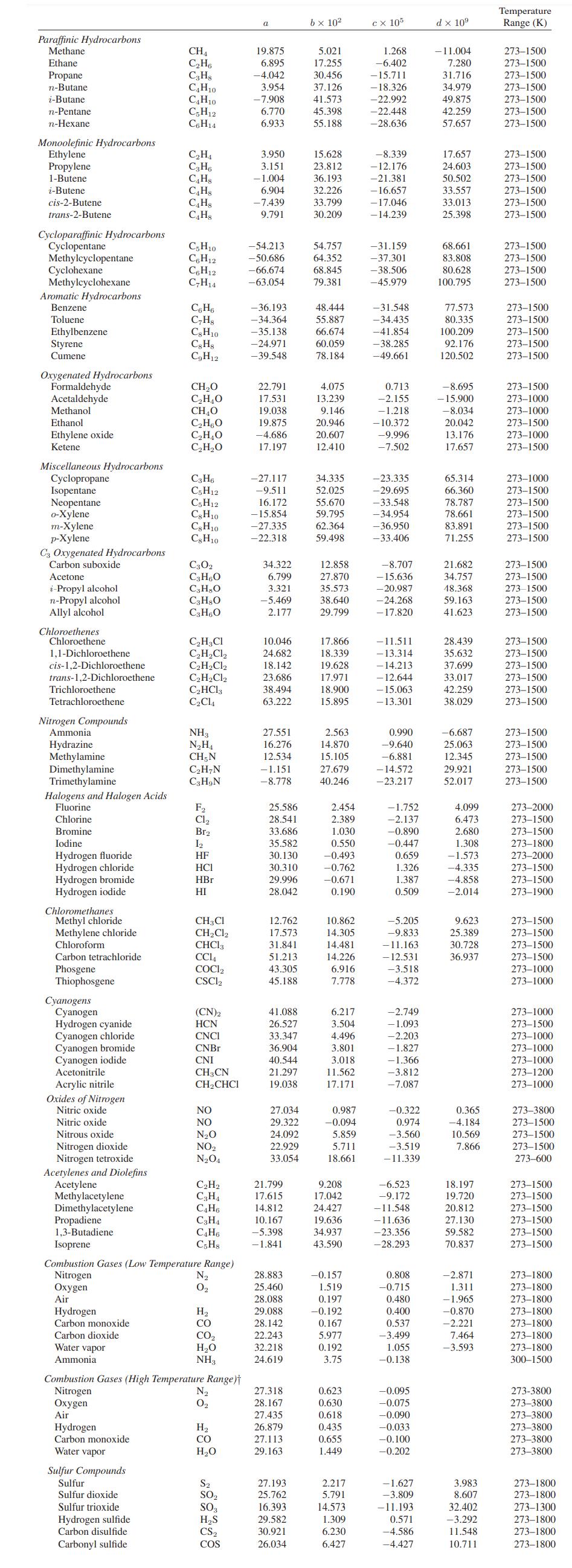
Paraffinic Hydrocarbons Methane Ethane Propane n-Butane i-Butane n-Pentane n-Hexane Monoolefinic Hydrocarbons Ethylene Propylene 1-Butene i-Butene cis-2-Butene trans-2-Butene Cycloparaffinic Hydrocarbons Cyclopentane Methylcyclopentane Cyclohexane Methylcyclohexane Aromatic Hydrocarbons Benzene Toluene Ethylbenzene Styrene Cumene Oxygenated Hydrocarbons Formaldehyde Acetaldehyde Methanol Ethanol Ethylene oxide Ketene Miscellaneous Hydrocarbons Cyclopropane Isopentane Neopentane o-Xylene m-Xylene p-Xylene C3 Oxygenated Hydrocarbons Carbon suboxide Acetone i-Propyl alcohol n-Propyl alcohol Allyl alcohol Chloroethenes Chloroethene 1,1-Dichloroethene cis-1,2-Dichloroethene trans-1,2-Dichloroethene Trichloroethene Tetrachloroethene Nitrogen Compounds Ammonia Hydrazine Methylamine Dimethylamine Trimethylamine Halogens and Halogen Acids Fluorine Chlorine Bromine Iodine Hydrogen fluoride Hydrogen chloride Hydrogen bromide Hydrogen iodide Chloromethanes Methyl chloride Methylene chloride Chloroform Carbon tetrachloride Phosgene Thiophosgene Cyanogens Cyanogen Hydrogen cyanide Cyanogen chloride Cyanogen bromide Cyanogen iodide Acetonitrile Acrylic nitrile Oxides of Nitrogen Nitric oxide Nitric oxide Nitrous oxide Nitrogen dioxide Nitrogen tetroxide Acetylenes and Diolefins Acetylene Methylacetylene Dimethylacetylene Hydrogen Carbon monoxide Carbon dioxide Water vapor Ammonia CH₁ C₂H6 C3H8 C4H10 C4H10 C5H12 C6H14 C₂H4 C3H6 C4H8 C4H8 C₂H8 C4H8 Sulfur Compounds Sulfur Sulfur dioxide Sulfur trioxide Hydrogen sulfide Carbon disulfide Carbonyl sulfide C5H10 C6H12 C6H12 C₂H14 C6H6 C7H8 Cg H10 Cg Hg C₂H12 CH₂O C₂H₂O CH₂O C₂H5O C₂H₂O C₂H₂O C3H6 C5H12 C5H12 C8H10 Cg H10 C8H10 C302 C3H6O C3H8O C3H8O C3H6O C₂H3C1 C₂H₂Cl₂ C₂H₂Cl₂ C₂H₂Cl₂ C₂HCl3 C₂C14 NH3 N₂H₁ CH, N C₂H7N C3H9N F₂ Cl₂ Br₂ 1₂ HF HCI HBr HI CH3 Cl CH₂Cl₂ CHC13 CC14 COCI₂ CSC12 (CN)2 HCN CNCI CNBr CNI CH3CN CH₂CHCI NO NO N₂O NO₂ N₂O4 Propadiene 1,3-Butadiene Isoprene Combustion Gases (Low Temperature Range) Nitrogen Oxygen Air C2H2 C3H₁ C4H6 C3H₁ C4H6 C5H8 N₂ 0₂ H₂ CO CO2 H₂O NH3 Combustion Gases (High Temperature Range) Nitrogen Oxygen Air Hydrogen. Carbon monoxide Water vapor N₂ 0₂ H₂ CO H₂O S₂ SO₂ SO3 H₂S CS₂ COS 19.875 6.895 -4.042 3.954 -7.908 6.770 6.933 3.950 3.151 -1.004 6.904 -7.439 9.791 -54.213 -50.686 -66.674 -63.054 -36.193 -34.364 -35.138 -24.971 -39.548 22.791 17.531 19.038 19.875 -4.686 17.197 -27.117 -9.511 16.172 -15.854 -27.335 -22.318 34.322 6.799 3.321 -5.469 2.177 10.046 24.682 18.142 23.686 38.494 63.222 27.551 16.276 12.534 -1.151 -8.778 25.586 28.541 33.686 35.582 30.130 30.310 29.996 28.042 12.762 17.573 31.841 51.213 43.305 45.188 41.088 26.527 33.347 36.904 40,544 21.297 19.038 27.034 29.322 24.092 22.929 33.054 21.799 17.615 14.812 10.167 -5.398 -1.841 28.883 25.460 28.088 29.088 28.142 22.243 32.218 24.619 27.318 28.167 27.435 26.879 27.113 29.163 27.193 25.762 16.393 29.582 30.921 26.034 b x 10² 5.021 17.255 30.456 37.126 41.573 45.398 55.188 15.628 23.812 36.193 32.226 33.799 30.209 54.757 64.352 68.845 79.381 48.444 55.887 66.674 60.059 78.184 4.075 13.239 9.146 20.946 20.607 12.410 34.335 52.025 55.670 59.795 62.364 59.498 12.858 27.870 35.573 38.640 29.799 17.866 18.339 19.628 17.971 18.900 15.895 2.563 14.870 15.105 27.679 40.246 2.454 2.389 1.030 0.550 -0.493 -0.762 -0.671 0.190 10.862 14.305 14.481 14.226 6.916 7.778 6.217 3.504 4.496 3.801 3.018 11.562 17.171 0.987 -0.094 5.859 5.711 18.661 9.208 17.042 24.427 19.636 34.937 43.590 -0.157 1.519 0.197 -0.192 0.167 5.977 0.192 3.75 0.623 0.630 0.618 0.435 0.655 1.449 2.217 5.791 14.573 1.309 6.230 6.427 cx 105 1.268 -6.402 - 15.711 -18.326 -22.992 -22.448 -28.636 -8.339 -12.176 -21.381 -16.657 - 17.046 -14.239 - 31.159 -37.301 -38.506 -45.979 -31.548 -34.435 -41.854 -38.285 -49.661 0.713 -2.155 -1.218 -10.372 -9.996 -7.502 -23.335 -29.695 -33.548 -34.954 -36.950 -33.406 -8.707 -15.636 -20.987 -24.268 - 17.820 -11.511 -13.314 -14.213 - 12.644 -15.063 -13.301 0.990 -9.640 -6.881 - 14.572 -23.217 -1.752 -2.137 -0.890 -0.447 0.659 1.326 1.387 0.509 -5.205 -9.833 -11.163 -12.531 -3.518 -4.372 -2.749 - 1.093 -2.203 -1.827 -1.366 -3.812 -7.087 -0.322 0.974 -3.560 -3.519 -11.339 -6.523 -9.172 -11.548 -11.636 -23.356 -28.293 0.808 -0.715 0.480 0.400 0.537 -3.499 1.055 -0.138 -0.095 -0.075 -0.090 -0.033 -0.100 -0.202 -1.627 -3.809 - 11.193 0.571 -4.586 -4.427 d x 10⁹ -11.004 7.280 31.716 34.979 49.875 42.259 57.657 17.657 24.603 50.502 33.557 33.013 25.398 68.661 83.808 80.628 100.795 77.573 80.335 100.209 92.176 120.502 -8.695 - 15.900 -8.034 20.042 13.176 17.657 65.314 66.360 78.787 78.661 83.891 71.255 21.682 34.757 48.368 59.163 41.623 28.439 35.632 37.699 33.017 42.259 38.029 -6.687 25.063 12.345 29.921 52.017 4.099 6.473 2.680 1.308 -1.573 -4.335 -4.858 -2.014 9.623 25.389 30.728 36.937 0.365 -4.184 10.569 7.866 18.197 19.720 20.812 27.130 59.582 70.837 -2.871 1.311 -1.965 -0.870 -2.221 7.464 -3.593 3.983 8.607 32.402 -3.292 11.548 10.711 Temperature Range (K) 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1000 273-1000 273-1500 273-1000 273-1500 273-1000 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-2000 273-1500 273-1500 273-1800 273-2000 273-1500 273-1500 273-1900 273-1500 273-1500 273-1500 273-1500 273-1000 273-1000 273-1000 273-1500 273-1000 273-1000 273-1000 273-1200 273-1000 273-3800 273-1500 273-1500 273-1500 273-600 273-1500 273-1500 273-1500 273-1500 273-1500 273-1500 273-1800 273-1800 273-1800 273-1800 273-1800 273-1800 273-1800 300-1500 273-3800 273-3800 273-3800 273-3800 273-3800 273-3800 273-1800 273-1800 273-1300 273-1800 273-1800 273-1800
Step by Step Answer:



Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Students also viewed these Engineering questions
-
An oil trader wants to buy 5,000 barrels of Brent Crude Oil but has the opportunity to buy just 1,000 barrels of Brent Crude Oil at a price of $65 per barrel if he also buys 2,000 barrels of West...
-
Redo Problem 6.22 with the SoaveRedlich-Kwong equation of state. Problem 6.22 A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming...
-
A spherical balloon has an initial diameter of 1 m and contains argon gas at 200 kPa, 40C. The balloon is connected by a valve to a 500-L rigid tank containing carbon dioxide at 100 kPa,...
-
Aussie Infonautics, Inc., produces handheld Windows CE¢-compatible organizers. Aussie Infonautics markets three different handheld models. PalmPro is a soupedup version for the executive on the...
-
Z sells cameras to A, B, C, and D for $110 per camera. Y, one of Zs competitors, sells a comparable camera to A for $101.50. Z, in response to this competitive pressure from Y, lowers its price to A...
-
For each of the agent types listed in Exercise 2.5, characterize the environment according to the properties given in Section 2.3, and select a suitable agent design. The following exercises all...
-
In 2004, Jui-Chen Lin, a citizen of China, entered into an agreement with Robert Chiu and Charles Cobb, citizens of the United States, to form an LLC to acquire and operate a fast-food restaurant in...
-
United Resources Company obtained a charter from the state in January 2011, which authorized 200,000 shares of common stock , $1 par value. During the first year, the company earned $475,000 and the...
-
A 47-kg pole vaulter running at 10 m/s vaults over the bar. Her speed when she is above the bar is 1.5 m/s. Neglect air resistance, as well as any energy absorbed by the pole, and determine her...
-
By measuring the temperature change and the specific volume change accompanying a small pressure change in a reversible adiabatic process, one can evaluate the derivative (SP) and the adiabatic...
-
Evaluate the difference for the ideal and van der Waals gases, and for a gas that obeys the virial equation of state. (3) - (0) P V
-
Divide your group into two smaller subgroups. Have group 1 follow the processes specified in this chapter for creating prototypes. Using a CASE tool or a word processor, group 1 should devise two...
-
Diego Company produces three styles of purses: style A, B, and C Style A Style B Style C Sales $210,000 $115,000 $60,000 Variable Expenses $64,000 $40,000 $40,000 Fixed Expenses $108,000 $60,000...
-
Bill Johnson's will provides that $10,000 a year would be paid to his widow and $5,000 a year to his son out of the estate's income. There were no charitable contributions made. If the estate's...
-
DEF Company declares and distributes a 30% common stock dividend when it has 60,000 shares of $10 par common stock outstanding. The market price per share is $75 at the date of declaration. Which...
-
How much will Haveford Company move out of finished goods for the month of March? Beginning balances on March 1st are below: Raw materials: $2,000 Work in process: $1,000 Finished goods: $9,000 In...
-
If the variable cost per unit to produce a skateboard is $15 and the contribution rate is 30%, what is the contribution margin?
-
What are the three cycles of transaction processing systems?
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
The following table shows how many hours in process reactors A and B are required to produce 1 ton each of chemical products 1, 2, and 3. The two reactors are available for 35 and 40 hrs per week,...
-
See Figure P13. Assume that no vehicles stop within the network. A traffic engineer wants to know if the traffic flows f 1 , f 2 , . . . , f 7 (in vehicles per hour) can be computed given the...
-
Use MATLAB to solve the following problem: x - 3y = 2 x + 5y = 18 4x - 6y = 20
-
Explain in simple terms why an energing technology is charecterised as fast growth?
-
Daphne, a sales representative, is placed in a group with an engineer from operations, a human resource specialist, and a financial manager to develop new uses for the company s existing products....
-
As we have reached the midpoint of our international business course, lets pause to teflect what are some ket concepts and why are thise concepts relavant?

Study smarter with the SolutionInn App


