Evaluate the difference for the ideal and van der Waals gases, and for a gas that obeys
Question:
Evaluate the difference
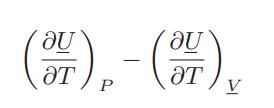
for the ideal and van der Waals gases, and for a gas that obeys the virial equation of state.
Transcribed Image Text:
(3²) - (0)₁ P V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The ideal gas equation is PV nRT where P is the pressure V is the volume n is the number of moles of gas R is the ideal gas constant T is the temperat...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted by 1.50 mol of SO 2 (g) when it is confined at 298 K to a volume of (a) 100.0 L; (b) 50.0 L; (c) 20.0...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
When they hear the name Credit Suisse, my grandchildren will likely have to google (or whichever search engine exists then) these two words. For most of my contemporaries here in Switzerland, Credit...
-
Universal Video sells $40 million worth of video recording equipment in the United States. The total sales of such equipment in the United States is $100 million. One-half of Universals sales is to...
-
Implement a simple reflex agent for the vacuum environment in Exercise 2.7. Run the environment simulator with this agent for all possible initial dirt configurations and agent locations. Record the...
-
Amanda Forbes was hired as a nail technician by Showmann, Inc., in 2011. In 2017, Forbes attended a work-related holiday party where Showmann distributed raffle tickets to employees. One of the...
-
Gravois, Inc., incurred the following costs during June: Selling expenses . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $158,375 Direct labor . . . . . . . . . . . . . . . . ....
-
A polar exploration team based near the south pole is faced with an emergency in which a very important housing and supplies module must be lifted by a special 1 crane, swung across a deep glacial...
-
A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming there is no heat transfer between the gas and the tank, find the pressure in...
-
For steam at 500 C and 10 MPa, using the Mollier diagram, a. Compute the Joule-Thomson coefficient = (T/P) H . b. Compute the coefficient S = (T/P) S . c. Relate the ratio ( H / S ) T /(H/S) P to ...
-
We want to bond together two slabs of a solid material, each with a thickness of 1.5 cm, to form a laminate by using a thin layer of a thermosetting glue, which fuses and forms a bond between the two...
-
Explain why do we call retained earnings "earned capital"? A Company was founded in 2013. Its yearly earnings and dividend payments are own here: 2013: Net income of $4,000, paid zero dividends 2014:...
-
The shareholders' equity section of Houston Corp. at December 31, Y3 was: Common shares, no par value; authorized 20,000 shares; issued and outstanding 10,000 shares $50,000 Retained earnings 200,000...
-
On January 1, 2021, Daniel Faust loaned $112,695 cash to Joyce Ladd in the form of a zero-interest-bearing note (face amount, $150,000). The note is to be repaid on December 31, 2023. The prevailing...
-
Rook Company sells video and board games. Below is last year's income statement for the board game segment: Sales $4,000,000 Variable expenses $2,600,000 Contribution margin $1,400,000 Fixed expenses...
-
Sam is an independent contractor to Quick-save, a computer service company. Sam sells the company's services and is compensated for every sale he makes. Quick-save decided to terminate its...
-
What are the elements of a system?
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
The concept of thermal resistance described in Problem 8 can be used to find the temperature distribution in the at square plate shown in Figure P9(a). Figure P9 The plates edges are insulated so...
-
Compute the double integral Note that the region of integration lies to the right of the line y = x. Use this fact and a MATLAB relational operator to eliminate values for which y > x. A = 2 (x +...
-
Compute the triple integral A = xe dxdy dz Jo Ji 0, 2.
-
A work operation consisting of three elements has been subjected to a stopwatch time study. The recorded observa- tions are shown in the following table. By union contract, the allowance time for the...
-
What are the key enzymatic players involved in the orchestration of DNA replication, and what roles do they serve in the process?
-
Compute the c 32 element in the matrix, C, by the following matrix addition: C = 2A + 3B. A = 8 4 -3 3 -2 -2 6 9 9 9 7 -2 B = 4 -2 -7 2 -6 2 9 -2 7 2 9 -9

Study smarter with the SolutionInn App


