The amino acid dl-alanine HCOO-CNH 2 H-CH 3 exists in the following four forms in solution Determine
Question:
The amino acid dl-alanine HCOO-CNH2H-CH3 exists in the following four forms in solution
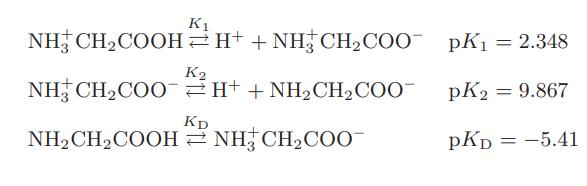
Determine the fraction of dl-alanine in each of these forms at
a. pH = 3
b. pH = 10
Transcribed Image Text:
K₁ NH CH₂COOH H+ + NHCH₂COO- NH CH,COO` =H*+NH,CH,COO KD NH₂ CH₂COOH NH3 CH₂COO- K2 pK₁ = 2.348 pK2 = 9.867 PKD = -5.41
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a At pH 3 the dlalanine will be mostly in the zwitterion form with a small amount in the cationic fo...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The OH group on the side chain of serine is not deprotonated at a pH of 12. However, the OH group on the side chain of tyrosine is deprotonated at a pH of 12. This can be verified by inspecting the...
-
Chi and Daniella are your clients and you are assessing their current financial situation and how it impacts their insurance needs. Chi and Daniella are married and they have a mortgage on their home...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Given that log 10 (x 2 y) = 4 and log 10 (x 4 /y 3 ) = 18, find the value of log 10 x and of log 10 y.
-
Biotechtron, Inc., has two research laboratories in the Midwest, one in Tulsa, Oklahoma, and one in Ames, Iowa. The owner of Biotechtron centralized the legal services function in the Tulsa office,...
-
McLean Company produced 2,500 units of product that required two standard hours per unit. The standard variable overhead cost per unit is $2.50 per hour. The actual variable factory overhead was...
-
The type of model building used by Pauling and by Watson and Crick involved the use of ball-and-stick units. Model building can now be done with computer software. Even though you may not be familiar...
-
The comparative statements of Beulah Company are presented below. Additional data: The common stock recently sold at $19.50 per share. Instructions Compute the following ratios for 2014. (a) Current....
-
Given that R is the relation defined on TL as follows: "For every m, n = Z, m R n 3|(m-n). (a) Prove that the relation is an equivalence relation. (b) Identify the distinct equivalence classes of...
-
Redo Illustration 13.5-2 including the ionization of water and including the effect of solution nonidealities. Illustration 13.5-2 A common analytical procedure is titration, in which a solution of a...
-
Ammonia, air and water is to be converted to a nitric acid solution. Ammonia is available as a liquid at 25C and its saturation pressure, air and water (as a liquid) are available at 1.013 bar and...
-
Why should a marketer be aware of competitors prices?
-
Imagine that you were a new CFO of Beily Inc., a childrens bicycle manufacturer. The president, Mr. Zhao, started the business 2 years ago. The firm manufactures two types of products, bicycles for...
-
Top Performance has been operating for several years in the outskirts of Ostersund, Sweden, and is a new manufacturer of a top-of-the-line outdoor jacket. You are starting an internship as assistant...
-
The debt and equity section of the TOTAL Group balance sheet is shown here (in million euros). The current market price of the common shares is 200. Reconstruct the financial statement assuming that...
-
The General Meeting of Remy Cointreau SA, listed on Euronext Paris, held in July 2015, set the overall dividend at 1.53 per share. The Meeting decided to grant every shareholder payment of the...
-
Door Company sells products in cash and in credit as follows: cash sales are 60 percent of total sales, and credit sales are 40 percent. 10 percent of the credit sales are collected in the month of...
-
Duval Co. issues four- year bonds with a $ 100,000 par value on June 1, 2013, at a price of $ 95,948. The annual contract rate is 7%, and interest is paid semiannually on November 30 and May 31. 1....
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
Draw the shear and moment diagrams for the beam. 6 kN/m 3 kN/m 3 m
-
Draw the shear and moment diagrams for the beam. 4 kN/m 2 kN/m B 3 m- 1.5 m
-
Draw the shear and moment diagrams for the beam. 3 kN 6 kN/m B 1.5 m 1.5 m
-
The prosecutor tried to convince the jury that the defendant was guilty because he owned a hammer that produced an impression similar to that found at the crime scene. As the defendants attorney, how...
-
A 3 MeV fast neutron hits an alpha-iron target (threshold energy 40 eV, atomic wt. 55.85) creating a PKA. Find out the number of displaced atoms per PKA using : a) simple K-P model, b) Snyder-Neufeld...
-
The Birth of a Companyand an industry On a cold February day in 1886, Charles Martin Hall filled a crucible with a cryolite bath containing alumina and passed an electric current through it. The...

Study smarter with the SolutionInn App


