When water and n-propanol are isothermally mixed, heat may be either absorbed (Q > 0) or evolved
Question:
When water and n-propanol are isothermally mixed, heat may be either absorbed (Q > 0) or evolved (Q
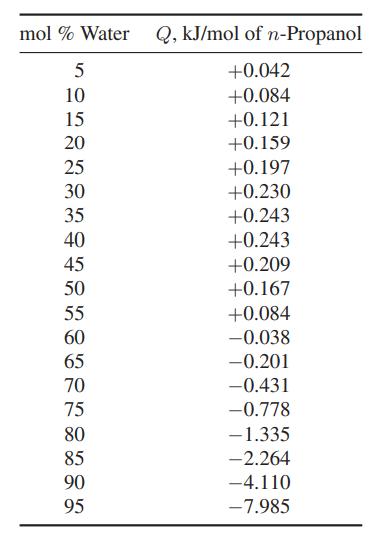
Plot (H̅W − HW) and (HNP − H̅NP) over the whole composition range.
Transcribed Image Text:
mol % Water Q, kJ/mol of n-Propanol 5 +0.042 10 +0.084 15 +0.121 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 +0.159 +0.197 +0.230 +0.243 +0.243 +0.209 +0.167 +0.084 -0.038 -0.201 -0.431 -0.778 -1.335 -2.264 -4.110 -7.985
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The molar integral heat of solution s H is defined as the change in enthalpy that results when 1 mole of solute (component 1) is isothermally mixed with N 2 moles of solvent (component 2) and is...
-
The following data are available for the isothermal heat of mixing of trichloromethane (1) and ethanol (2) at 30C [reference: J. P. Shatas, M. M. Abbott, and H. C. Van Ness, J. Chem. Eng. Data, 20,...
-
Use the data in problem 8.29 to compute the partial molar enthalpies of pyridine and acetic acid in their mixtures at 25C over the whole composition range. Problem 8.29 Two streams containing...
-
The data in the chart below is for the distance (in cm) to the near point, the point nearest the eye at which the eye can accurately focus, at a persons age. Age (years) Near Point (cm) 10 7.5 20 9...
-
New West Fruit Corporation (New West) and Coastal Berry Corporation are both brokers of fresh strawberries. In the second half of 2009, New Wests predecessor, Moncs Consolidated Produce, Inc., loaned...
-
1. Assume an economy of two firms and two consumers. The two firms pollute. Firm one has a marginal savings function of MS, (e) = 5 - e where e is the quantity of emis- sions from the firm. Firm two...
-
James Lewis, a resident of Kentucky, sustained an injury while operating a Caterpillar bulldozer. He filed suit against Caterpillar, a company incorporated in Delaware but with its principal place of...
-
Effects of operating leverage on profitability Webster Training Services (WTS) provides instruction on the use of computer software for the employees of its corporate clients. It offers courses in...
-
Find an application of OLAP (online analytical processing) for FIU. How can FIU use multidimensional analytical queries to generate BI? Some examples?
-
The heat-of-mixing data of Featherstone and Dickinson [J. Chem. Thermodyn., 9, 75 (1977)] for the n-octanol + n-decane liquid mixture at atmospheric pressure is approximately fit by with T in K and x...
-
Mattingley and Fenby [J. Chem. Thermodyn. 7, 307 (1975)] have reported that the enthalpies of triethylamine-benzene solutions at 298.15 K are given by where x B is the mole fraction of benzene and H...
-
An ideal gas undergoes a process between two specified temperatures, first at constant pressure and then at constant volume. For which case will the ideal gas experience a larger entropy change?...
-
Explain the characteristics of ethical leaders. Distinguish between types of leadership. Describe what ethical leadership looks like in the accounting profession. Describe the components of...
-
Need a good solid explanation on ethical leadership and some gray area that can be posed by it. Like in what case ethical leadership can lead to a poor decisions over common sense. ?
-
Describe voter ID laws in a state of your choosing. Summarizeany recent developments or controversies regarding voter ID laws inthe state you have chosen. Analyze and describe the pros and cons on...
-
Explain one of the 10 sections from "The World Delivered to Your Home" (from Anders' The Obsolescence of Human Beings). First, state the main claim. Then, explain the claim fully. Give contemporary...
-
Explain the 5 different phases for new pizza restaurant project: Phase 1 - Define Phase 2 - Measure Phase 3 - Analyze Phase 4 - Improve Phase 5 - Control Name at least two Six Sigma opportunities for...
-
Distinguish between the value chain, the supply chain, and the distribution chain.
-
The cash records of Holly Company show the following four situations. 1. The June 30 bank reconciliation indicated that deposits in transit total $720. During July, the general ledger account Cash...
-
In 1925, the Try-State Tornado ripped a 219-mi path of destruction through Missouri, Illinois, and Indiana, killing a record 695 people. The maximum winds in the tornado were 318 mph. Express the...
-
Uphill water slides are becoming more popular at large water parks. Uphill speeds of riders can reach 19 ft/s. Express this speed in mph.
-
A brand-new engineering hire is late for her first product development team meeting. She gets out of her car and starts running 8 mph. It is exactly 7:58 a.m., and the meeting starts at exactly 8:00...
-
Bond J has a coupon rate of 6 percent and Bond K has a coupon rate of 12 percent. Both bonds have 15 years to maturity, make semiannual payments, and have a YTM of 9 percent. a. b. If interest rates...
-
Rebel Gear Corporation (Rebel) is a clothing manufacturer and distributor. The company specializes in college-branded clothing and jewelry. On August 1, 2022, Rebel entered into an agreement with...
-
a) A man borrows 2000 for 160 days at 6,50%. What amount must he repay? (use a 360-days year) b) Use the simple interest formula to find the rate on a 2000 loan for which the interest was 12 for 30...

Study smarter with the SolutionInn App


