From the CV below, estimate the formal potential for the redox couple. Anodic current Cathodic current 5.0
Question:
From the CV below, estimate the formal potential for the redox couple.
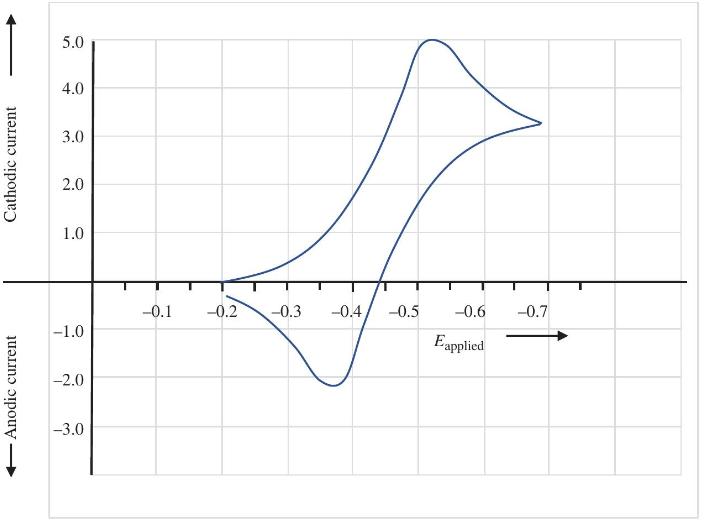
Transcribed Image Text:
Anodic current Cathodic current 5.0 4.0 3.0 2.0 1.0 -0.1 -0.2 -0.3 -0.4 -0.5 -0.6 -0.7 -1.0 Eapplied -2.0 -3.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Anodic Current Cathodic ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Electroanalytical Chemistry Principles Best Practices And Case Studies
ISBN: 9781119538592,9781119538585
1st Edition
Authors: Gary A. Mabbott
Question Posted:
Students also viewed these Engineering questions
-
If the experiment in Problem 5.3 was recorded using a saturated calomel reference electrode at \(25^{\circ} \mathrm{C}\), what is the value of \(\mathrm{E}^{\circ \prime}\) for the redox couple on...
-
Does the voltammogram in Problem 5.3, indicate any possibility that the reduced or the oxidized form is unstable under the conditions in this solution? Support your answer with a calculation. Data...
-
For the voltammogram in Problem 5.3, determine whether the one-electron transfer process is reversible in the electrochemical sense. Data from Problem 5.3 From the CV below, estimate the formal...
-
4. This problem investigates nearest neighbor and bilinear interpolation. For simplicity, we will focus on estimating the image intensity at a single location. Interpolation is used when transforming...
-
What drivers of cost advantage make Walmart such a successful organization in the world of low-cost leadership?
-
Water is to be cooled at the rate of \(10 \mathrm{~kg} / \mathrm{s}\) from \(317 \mathrm{~K}\) to \(300 \mathrm{~K}\) in a countercurrent cooling tower under conditions such that \(H_{t O G}=2.5...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
Devine Brown opened Devines Carpet Cleaners on March 1. During March, the following transactions were completed. Mar. 1 Invested $10,000 cash in the business. 1 Purchased used truck for $6,000,...
-
3) The cost and revenue function in dollar terms for a fast-food joint is given as follows: C(x)=200x+200 and R(x) = 50x X + 4 If x is measured in hundreds of units, what is the break-even point for...
-
If for a cyclic voltammogram the \(E^{\mathrm{O} \prime}\) for quinone, \(Q\), appears at a \(+0.220 \mathrm{~V}\) at a platinum electrode (versus silver/silver chloride ref. electrode) at...
-
A \(33.6 \mathrm{mg}\) sample of a freeze-dried enzyme preparation was injected into a Karl Fisher coulometric titrator and the endpoint was reached in 27.6 seconds at a current level of \(150.7...
-
Suppose there are three objectives, not just two: the total advertising cost, the total number of excess exposures to men, and the total number of excess exposures to women. Continuing the approach...
-
Mary had a large investment portfolio consisting of equities that she has held for many years. The securities appreciated significantly in value over the years. When she first bought these equities...
-
During preparation of your bid, you learn that there is local noise ordinance that limits the work hours due to limits to construction noise. What should we do?
-
The Heather Company's fiscal year ends on June 30.Its employees (with at least three months of experience) are entitled to 12 paid sick days annually for each calendar year beginning on January 1.An...
-
Name two actuarial assumptions that may be considered in valuing a defined benefit plan and explain why they are needed?
-
Justin(Dependent) Income from working as an amateur coder $1,950 Income working as a golf caddy $4,000 Dividend Income $2,350 Interest Income $750, What is his taxable income? Does he have a filing...
-
Refrigerant 22vapor enters the compressor of a refrigeration system at an absolute pressure of 20lbf/in2 a pressure gage at the compressor exit indicates a pressure of 280lbf/in2 (Gage) the...
-
Explain what is meant by vicarious liability and when it is available?
-
Kinetic data for the reaction between sulfuric acid and diethyl sulfate are given in Examples 6.1 and 6.4. Suppose that V = 25.4 L, q Af = q Bf , c Af = 11.0 g-mol/L, c Bf = 5.5 g-mol/L, and the...
-
The formation of sugars from biomass is of considerable interest in a variety of applications, including energy alternatives to fossil fuel. Harris and Kline studied the formation of glucose from...
-
Consider the process in Example 13.4. Can you reduce the required area substantially by changing the coolant flow rate? Example 13.4 We must reduce the temperature of a small process steam flowing at...
-
In Europe, the floor of a building at ground level is called the ground floor. What is called the second floor in America is called the first floor in Europe, and so on. (a) If an elevator in a tall...
-
What role do social networks and interpersonal relationships play in both perpetuating and challenging existing social structures ?
-
A homeowner is thinking about buying an electric heat pump to save on heating costs. The heat pump is said to reduce fuel consumption by 10 MMBtu per year for 20 years. It costs $1,400 upfront and...

Study smarter with the SolutionInn App


