Balance the following skeletal equations and classify them as acidbase or redox: (a) MgC (s) + HO(1)CH(g)
Question:
Balance the following skeletal equations and classify them as acid–base or redox: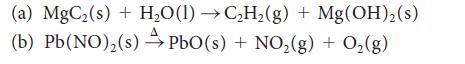
Transcribed Image Text:
(a) MgC₂ (s) + H₂O(1)→C₂H₂(g) + Mg(OH)₂(s) (b) Pb(NO)₂(s) PbO (s) + NO₂(g) + O₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a MgCs 2 HO1 ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Balance the following skeletal equations and classify them as acidbase or redox: (a) CH4(g) + Sg(s) CS (1)+ HS(g) (b) Sn(s) + KOH(aq) + HO(1)KSn(OH),(aq) + H(g)
-
Complete and balance the equations below, and classify them as precipitation, acidbase, gasforming, or oxidationreduction reactions. Show states for reactants and products (s, , g, aq). (a) NiCO3 +...
-
Balance each of the following skeletal equations by using oxidation and reduction half-reactions. All the reactions take place in acidic solution. Identify the oxidizing agent and reducing agent in...
-
Please help I am confused and really stuck here and can't properly answer this one. Does it meet all the assumptions? b. Do the data meet all the assumptions of the appropriate statistical test...
-
Compost Science, Inc. (CSI), is in the business of converting Bostons sewage sludge into fertilizer. The business is not in itself very profitable. However, to induce CSI to remain in business, the...
-
Ramos Corporation is considering the elimination of one of its segments. The segment incurs the following fixed costs. If the segment is eliminated, the building it uses will be sold. Advertising...
-
A stock with current value \(S(0)=100\) has an expected growth rate of its logarithm of \(u=12 \%\) and a volatility of that growth rate of \(\sigma=20 \%\). Find suitable parameters of a binomial...
-
Midland Oil has $1,000 par value bonds outstanding at 8 percent interest. The bonds will mature in 25 years. Compute the current price of the bonds if the present yield to maturity is: a. 7% b. 10%...
-
The comparative financial statements prepared at December 31, 2015, for Prince Company showed the following summarized data: 2015 2014 Income statement: Sales revenue $190,200* $168,600 Cost of goods...
-
Lead azide, Pb(N 3 ) 2 , is used as a detonator. (a) What volume of nitrogen at STP (1 atm, 0 C) does 1.5 g of lead azide produce when it decomposes into lead metal and nitrogen gas? (b) Would 1.5 g...
-
(a) Arrange the chlorine oxoacids in order of increasing oxidizing strength. (b) Suggest an interpretation of that order in terms of oxidation numbers.
-
McDonalds restaurants do the bulk of their business at lunchtime, but have found that promotionally-priced meals at breakfast and dinner make a significant profit contribution. Does the success of...
-
16 9 9 J n 20 23 17 26 27 1 30 33 1 24 18 21 Question 31 (12 points) On July 31, Bristlecone Pine Art Ltd. had a $64,900 balance in Accounts Receivable and a $4,300 credit balance in Allowance for...
-
You were hired to be the digital marketing strategist for Patricia Nash Handbags. https://patricianashdesigns.com/ You can find all the data you need in the final exam Google Drive folder. This data...
-
QUESTION 3: ACCOUNTING FOR INCOME TAX The accounting profit after tax for Colesworths Ltd for the year ended 30 June 2023 was $2,089,500 and included the following revenue and expense items:...
-
Analyze and compare Amazon.com, Best Buy, and Wal-Mart The condensed income statements through operating income for Amazon.com, Inc. (AMZN), Best Buy Co., Inc. (BBY), and Wal-Mart Stores, Inc. (WMT),...
-
3. Given below are selected transactions for January for Leah Garcia, an interior designer: Jan. Invested P200,000 cash to start the business. 2 3 5 Paid P6,000 cash for advertising and P10,000 for...
-
How could a CPA firm use mix variances to evaluate performance?
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
The ionization energy of CO is greater than that of NO. Explain this difference based on the electron configuration of these two molecules.
-
A HartreeFock calculation using the minimal basis set of the 1s, 2s, 2p x , 2p y , and 2p z AOs on each of N and O generated the energy eigenvalues and AO coefficients listed in the following table....
-
Calculate the value for the coefficients of the AOs in Example Problem 23.4 for S 12 = 0.45. How are they different from the values calculated in that problem for S 12 = 0.3? Can you offer an...
-
Compose an email with link to book now for an online travel agency.
-
Evaluate the article: Boehm, Kunze, F., & Bruch, H. (2014). Spotlight on Age-Diversity Climate: The Impact of Age-Inclusive HR Practices on Firm-Level Outcomes. Personnel Psychology , 67 (3),...
-
Carbonate-bicarbonate buffer is extensively used in molecular and cell biology, biochemistry and in the medical field. It can be prepared by mixing aqueous solutions of Na2CO3 and NaHCO3. CO2 (aq) +...

Study smarter with the SolutionInn App


