Balance the following skeletal equations and classify them as acidbase or redox: (a) CH4(g) + Sg(s)
Question:
Balance the following skeletal equations and classify them as acid–base or redox: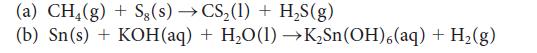
Transcribed Image Text:
(a) CH4(g) + Sg(s) → CS₂ (1)+ H₂S(g) (b) Sn(s) + KOH(aq) + H₂O(1)→K₂Sn(OH),(aq) + H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The first equation is CHg Ss CSl HSg To balance this equationwe need to do the following Balance the ...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Balance the following skeletal equations and classify them as acidbase or redox: (a) MgC (s) + HO(1)CH(g) + Mg(OH)(s) (b) Pb(NO)(s) PbO (s) + NO(g) + O(g)
-
Complete and balance the equations below, and classify them as precipitation, acidbase, gasforming, or oxidationreduction reactions. Show states for reactants and products (s, , g, aq). (a) NiCO3 +...
-
Balance each of the following skeletal equations by using oxidation and reduction half-reactions. All the reactions take place in acidic solution. Identify the oxidizing agent and reducing agent in...
-
Is the answer correct? The graph of a y = f(x) is shown. 6 + 5 14 m ~ 1 -6 -5 -4 -3 2 -1 -1 -2 -3 A w -4 -5- -6+ 1 2 3 4 The domain of f(x) is x = [-3,3] The domain of g(x) = notation) S 6 f(x) is a ...
-
In March 2001, Fly Papers stock sold for about $73. Security analysts were forecasting a long-term earnings growth rate of 8.5 percent. The company was paying dividends of $1.68 per share. a. Assume...
-
Allard Manufacturing Company established the following standard price and cost data. Sales price ............... $10.00 per unit Variable manufacturing cost ........ $6 per unit Fixed manufacturing...
-
The floating rate portion of a plain vanilla interest rate swap with yearly payments and a notional principal of one unit has cash flows at the end of each year defining a stream starting at time 1...
-
Public transportation and the automobile are two methods an employee can use to get to work each day. Samples of times recorded for each method are shown. Times are in minutes. a. Compute the sample...
-
Chong, Inc. provides advice to art enthusiast interested in purchasing art at Sotheby's annual art auctions. On December 31, 2012, Chong's account balances are shown below in the Adjusted Trial...
-
Write the chemical equation for the reaction between (a) Cesium and oxygen (cesium reacts with oxygen in the same way as potassium); (b) Sodium oxide and water; (c) Lithium and hydrochloric acid; (d)...
-
The diatomic molecule BF can be obtained by the reaction between BF 3 and B at a high temperature and low pressure. (a) Identify the electron configuration of the molecule in terms of the occupied...
-
1. Think of some advertisements which deliberately seek to make demand less elastic. 2. Imagine that Sunshine sunflower spread, a well-known brand, is advertised with the slogan: It helps you live...
-
On 1 October 2022 Pally Co acquired 90% of the ordinary shares of Sally Co by issuing 100 million shares at an agreed value of $1.60 per share and $140 million in cash. At that time (1.10.2022), the...
-
Question 2 (15 points) Saved Using Lenovo as a reference organisation i. Identify 3 sales divisions ii. Identify 2 sales channels [5 marks] [5 marks] iii. Draw an organisation structural diagram...
-
Figure Q2 shows an impulsive acceleration having duration 0.02 seconds and maximum magnitude of 10 m/sec. 10 accn. [m/sec] 0.2 0.4 0.6 0.8 1 time [seconds] Figure Q2 a) If a single degree of freedom...
-
Chase Company uses the perpetual inventory method. The inventory records for Chase reflected the following information: January 1 Beginning inventory January 12 Purchase January 18 Sales January 21...
-
PART I. ON THE ANSWER SHEET TYPE THE CORRECT ANSWER AS "TRUE" OR "FALSE" NEXT TO THE NUMBER OF THE TEST ITEM. 1. In every deductive argument the premises are linked. 2. In every valid deductive...
-
If the returns from a security were known with certainty, what shape would the probability distribution of returns graph have?
-
Vince, Inc. has developed and patented a new laser disc reading device that will be marketed internationally. Which of the following factors should Vince consider in pricing the device? I. Quality of...
-
Why are localized and delocalized models equally valid for describing bonding in closed-shell molecules? Why cant experiments distinguish between these models?
-
The hybridization model assumes that atomic orbitals are recombined to prepare directed orbitals that have the bond angles appropriate for a given molecule. What aspects of the model can be tested by...
-
Why cant localized orbitals be represented in an MO energy diagram?
-
Oriole Company, a machinery dealer, leased a machine to Pharoah Corporation on January 1 , 2 0 2 5 . The lease is for an 8 - year period and requires equal annual payments of $ 3 3 , 3 0 0 at the...
-
Jorge and Anita, married taxpayers, earn $ 1 5 0 , 0 0 0 in taxable income and $ 4 0 , 0 0 0 in interest from an investment in City of Heflin bonds. ( Use the U . S . tax rate schedule for married...
-
The following information is available for ADT Company, which produces special - order security products and uses a job order costing system. Overhead is applied using a predetermined overhead rate...

Study smarter with the SolutionInn App


