Describe the cell based on the following half-reactions: where VO+ + 2H+ + e Zn+ +2e= VO+
Question:
Describe the cell based on the following half-reactions:
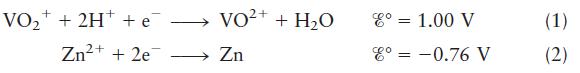
where
![T = 25C [VO+] = 2.0 M [H] = 0.50 M [VO+] = 1.0 x 10- M [Zn+] = 1.0 10- M](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1706/0/8/9/95565b0dde3d628f1706089955983.jpg)
Transcribed Image Text:
VO+ + 2H+ + e Zn+ +2e= VO+ + HO 2+ Zn 8 = 1.00 V 8 = -0.76 V (1) (2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The balanced cell reaction is obtained by reversing reaction 2 and multiplyi...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Consider the standard galvanic cell based on the following half reactions Cu2+ + 2e- Cu Ag+ + e- Ag The electrodes in this cell are Ag(s) and Cu(s). Does the cell potential increase, decrease, or...
-
The Stand-Alone Project requires you to perform an in-depth strategic audit of American Airlines, Inc. These past several years have been troubling times for the airline industry. Jet fuel prices...
-
Modify Index to make a program IndexByKeyword that takes a file name from the command line and makes an index from standard input using only the keywords in that file. Note: Using the same file for...
-
A train moves along the tracks at a constant speed v. A woman on the train throws a ball of mass m straight ahead with a speed v with respect to herself. (a) What is the kinetic energy gain of the...
-
An output interface in a switch is designed using the leaky bucket algorithm to send 8000 bytes/s (tick). If the following frames are received in sequence, show the frames that are sent during each...
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
The 2015 income statement and comparative balance sheet of Rolling Hills, Inc. follow: Additionally, Rolling Hills purchased land of $ 23,600 by financing it 100% with long- term notes payable during...
-
You are a co-op student at Modus Biosystems and have been asked to research whether there are ways to save money on their legal liability insurance. Modus currently pays $75,000 a year for a policy...
-
For the oxidationreduction reaction the appropriate half-reactions are Balance the redox reaction and calculate and K (at 25C). S406 (aq) + Cr+ (aq) 3+ Cr+ (aq) + S03 (aq)
-
For the cell reaction predict whether cell will be larger or smaller than cell for the following cases. 2Al(s) + 3Mn+ (aq) 2A1+ (aq) + 3Mn(s) Ecell = 0.48 V
-
How does the J-curve effect relate to the time path of currency depreciation?
-
In the paper market, the demand for paper decreases. The table above shows the new market paper demand schedule. The market is perfectly competitive and each company has the costs shown in the table...
-
1. Fix the below program that uses a function show_letter_grade to display a user's letter grade. 2. The user is prompted to enter their numeric grade in the function. # Activity 2.7 # This program...
-
Provide a calculation example for each of the following: o Patient's co-payment + insurance portion = full reimbursement o Patient's co-insurance + insurance portion = full reimbursement
-
Irene purchased an annuity with a 5000 premium payment which she deducted from her income tax the values in her contract will accumulate tax-deferred based only on this information what kind of...
-
5. An AVL tree is a binary tree in which at each vertex the heights of the right and left subtrees of that vertex differ by at most 1. If the AVL tree is of height h what is the maximum possible...
-
A friend comes to you with the following problem. I provided my boss a cost equation using regression analysis. He was unhappy with the results. He told me to do more work and not return until I had...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Many reactions that take place in water have analogous reactions in liquid ammonia (normal boiling point, 33C). (a)Write the chemical equation for the autoprotolysis of NH 3 . (b)Write the formulas...
-
The pH of several solutions was measured in a hospital laboratory; convert each of the following pH values into the molar concentration of H 3 O + ions: (a) 4.8 (the pH of a urine sample); (b) 0.7...
-
Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength: HCOOH, (CH 3 ) 3 NH + , N 2 H 5 + , HF. TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0 X 10-...
-
1. (10) If x is greater than or equal to zero, then assign the square root of x to the variable sqrt_x and print out the result. Otherwise, print out an error message about the argument of the square...
-
A car is accelerated from velocity v1 to v2 in the time period of t . The mass of the car is m. The inclined angle of the hill is a. The distance travelled along the hill is L. The vertical distance...
-
The predicted boundary pressure at the end of each specified time period is given below: Reservoir Aquifer Radius, ft h, f 2000 8 25 30 k, md 100 200 .% 15 22 .cp 2 0.8 Ca psi 1106 0.7106 Cpsi 2x106...

Study smarter with the SolutionInn App


