Predict the products of each of the following reactions and then balance each equation: (a) Mg (OH)
Question:
Predict the products of each of the following reactions and then balance each equation: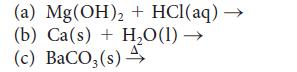
Transcribed Image Text:
(a) Mg (OH)₂ + HCl(aq) — (b) Ca(s) + H₂O(1)→ (c) BaCO3(s) 4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a MgOHs 2 HClaq ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Predict the products of each of the following reactions and then balance each equation: (a) Mg(s) + Br(1) (b) BaO(s) + Al(s) (c) CaO(s) + SiO (s)
-
Predict the products of each of the following reactions. (a) (b) (c) (d) OH Cl pyridine OH (1) NaH (2) CH2l HBr OH HNOg, H2SO4 H3C
-
Predict the major products of each of the following reactions and then balance the equations: (a) FeCrO4(s) + C(s) A (b) CrO (s) + HO*(aq) (c) MnO (s) + Al(s) A
-
Types of audit procedures Audit procedures are used to gatherevidence to support the auditors conclusions on the fairpresentation of a companys financial statements. Procedures can beperformed...
-
Look again at Table 4.7. a. How do free cash flow and present value change if asset growth rate is only 15 percent in years 1 to 5? If value declines, explain why. b. Suppose the business is a...
-
Rimes Company makes and sells lawn mowers for which it currently makes the engines. It has an opportunity to purchase the engines from a reliable manufacturer. The annual costs of making the engines...
-
Suppose that \(v_{1}, v_{2}, \ldots, v_{n}\) are positive numbers. The arithmetic mean and the geometric mean of these numbers are, respectively, (a) It is always true that \(v_{A} \geq v_{G}\)....
-
Miranda, Inc., manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2011 and 2010 are provided below. Selected missing...
-
The comparative financial statements prepared at December 31, 2015, for Prince Company showed the following summarized data: 2015 2014 Income statement: Sales revenue $190,200* $168,600 Cost of goods...
-
Suggest a Lewis structure for B 4 H 10 and deduce the formal charges on the atoms. There are four BHB bridges.
-
Lead azide, Pb(N 3 ) 2 , is used as a detonator. (a) What volume of nitrogen at STP (1 atm, 0 C) does 1.5 g of lead azide produce when it decomposes into lead metal and nitrogen gas? (b) Would 1.5 g...
-
The energy eigenstates of the infinite square well are of course not unique as a basis for the Hilbert space, which has no support at the boundaries of the well, where \(x=0\) and \(x=a\). For...
-
The complex electric field of a time harmonic field in free space is given by X E = a 100 (1 + j) e-j(2/5) x Assuming the distance x is measured in meters, find the: (a) (4 points) wavelength in...
-
Electronics Incorporated purchased 120 computers from its supplier on credit at a cost of $400 per computer (total cost of $48,000). The computers were purchased to be held for sale to customers...
-
A 20 kVA, 1000 V/200 V, 50 Hz single-phase transformer gave the following test results:- Open Circuit Test: 200 V, 3.5 A, 400 W on L.V. side Measurement Short Circuit Test: 50 V, 30 A, 1200 W on H....
-
CO1: Create communication documents using assigned software applications. CO2: Build training presentations using multiple application software. The scenario: Because the company is growing and...
-
Calculate the required return (weighted average cost of capital) Debt Equity Market Values Required return (weighted average cost of capital).. Calculate the Economic Profit (Economic Value Added)...
-
What are the advantages of using economic value added (EVA1) instead of ROI?
-
Evenflow Power Co. is considering a new project that is a little riskier than the current operations of the company. Thus, management has decided to add an additional 1.5% to the company's overall...
-
Using ζ as a variational parameter in the normalized function allows one to vary the size of the orbital. Show this by calculating the probability of finding the electron inside a sphere...
-
The overlap integral for Ï g and Ï u as defined in Section 23.3 is given by Plot S ab as a function of R/a 0 for ζ = 0.8, 1.0, and 1.2. Estimate the value of R/a 0 for which S...
-
Sketch out a molecular orbital energy diagram for CO and place the electrons in the levels appropriate for the ground state. The AO ionization energies are O2s: 32.3 eV; O2p: 15.8 eV; C2s: 19.4 eV;...
-
According to Percival and Lee (2022), what are the key barriers and catalysts to collective mobilization in the creative industries? Name each barrier and catalysator and explain how they hinder or...
-
Explain the role of social media in marketing and discuss the implications of social media for marketers.
-
Explain Netflix's early years and its transition from DVD sales to DVD rentals. Lastly, what were the results and experiences or lessons learned by Netflix from this change?

Study smarter with the SolutionInn App


