Predict the products of each of the following reactions and then balance each equation: (a) Mg(s) +
Question:
Predict the products of each of the following reactions and then balance each equation: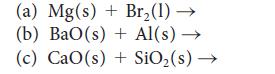
Transcribed Image Text:
(a) Mg(s) + Br₂(1)→ (b) BaO(s) + Al(s)→ (c) CaO(s) + SiO₂ (s) →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Lets predict the products and balance the chemical equa...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Predict the products of each of the following reactions and then balance each equation: (a) Mg (OH) + HCl(aq) (b) Ca(s) + HO(1) (c) BaCO3(s) 4
-
Predict the products of each of the following reactions. (a) (b) (c) (d) OH Cl pyridine OH (1) NaH (2) CH2l HBr OH HNOg, H2SO4 H3C
-
Predict the major products of each of the following reactions and then balance the equations: (a) FeCrO4(s) + C(s) A (b) CrO (s) + HO*(aq) (c) MnO (s) + Al(s) A
-
If a restaurant's total sales on a given day were $4,350.00, and the restaurant had served 365 customers, what is the average dollar sale? $10.05 $11.92 $11.35 $11.05
-
Consider the following three stocks: a. Stock A is expected to provide a dividend of $10 a share forever. b. Stock B is expected to pay a dividend of $5 next year. Thereafter, dividend growth is...
-
The accountant for Ericas Dress Shop prepared the following cash budget. Ericas desires to maintain a cash cushion of $20,000 at the end of each month. Funds are assumed to be borrowed and repaid on...
-
Derive the mean-variance hedge formula given by (12.12). h FT-Fo 2r var(FT) cov(x, FT) var(FT) (12.12)
-
Curtiss Construction Company, Inc., entered into a fixed-price contract with Axelrod Associates on July 1, 2011, to construct a four-story office building. At that time, Curtiss estimated that it...
-
Below are extracted information for XYZ Limited for its first - year account ending 3 1 December 2 0 2 1 : Equipment ( NCA ) 2 0 , 0 0 0 Utilities Expense 2 , 1 0 0 Cash 5 , 0 0 0 Accounts Receivable...
-
Arrange the following atoms in order of increasing first ionization energy: boron, thallium, gallium.
-
Write the chemical equation for the reaction between (a) Cesium and oxygen (cesium reacts with oxygen in the same way as potassium); (b) Sodium oxide and water; (c) Lithium and hydrochloric acid; (d)...
-
Subsidiarys inventory at the year-end included 180,000 purchased from its parent. Further goods invoiced by the parent at 45,000 were in transit. The parent invoices the subsidiary at cost plus 20%....
-
1. The following shows the demands and marginal revenue in two markets (D1 and MR1, and D2 and MR2) for a price discriminating firm along with total demand, DT, marginal revenue, MRT, and marginal...
-
You are a young design engineer working for a consulting firm. You have been asked to develop a specification for the installation and construction of a tall sign for a local Starbucks. The sign will...
-
Step 1 1 : Choose Apple Choose an organization that does marketing. Use the guidelines below as you make your selection. Make it easy on yourself and choose an organization you re interested in , ,...
-
Q2. Bank Run (30 points) Assume that there is an economy populated by a continuum [0,1] of households living in three periods, 0, 1, and 2. In period 0, each household is endowed with one unit of...
-
4.1 Each person's employment status is described in the following table. Based on the criteria used by the Bureau of Labor Statistics (BLS), indicate each person's status as Employed (E), Unemployed...
-
Under what conditions will a bonds current yield be equal to its yield to maturity?
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
How do the values of the AO coefficients in a MO differ for a delocalized and a localized bond?
-
What experimental evidence can you cite in support of the hypothesis that the electronegativity of a hybridized atom increases with increasing s-character?
-
Explain why all possible wave functions between the fully bonding and the fully anti-bonding are possible for the bands shown in Figure 24.22. Figure 24.22 N atomic orbitals 48 3.22B 28 2 atoms, N...
-
Write up an Essay on Search Engine Marketing (SEM) and Search Engine Optimization (SEO) in general. Then about SEO and SEM in the fashion apparel Industry.
-
Lui, Montavo, and Johnson plan to liquidate their Premium Pool and Spa business. They have always shared profit and losses in a 1 : 4 : 5 ratio, and on the day of the liquidation their balance sheet...
-
Company A has current sales of $ 1 1 , 5 7 9 , 8 6 2 and a 4 3 % contribution margin. Its fixed costs are $ 2 , 7 2 1 , 7 2 2 . Company B is a service firm with current service revenue of $ 6 , 9 6 3...

Study smarter with the SolutionInn App


