Write the half-reactions and devise a galvanic cell (write a cell diagram) to study each of the
Question:
Write the half-reactions and devise a galvanic cell (write a cell diagram) to study each of the following reactions: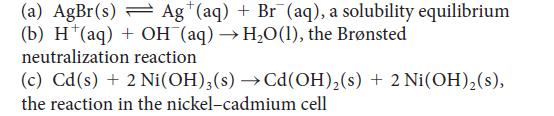
Transcribed Image Text:
(a) AgBr(s) Ag (aq) + Br (aq), a solubility equilibrium (b) H¹(aq) + OH (aq) →H₂O(1), the Brønsted neutralization reaction (c) Cd(s) + 2 Ni(OH)3(s) →Cd(OH)₂ (s)+ 2 Ni(OH)₂ (s), the reaction in the nickel-cadmium cell
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a anode Ags Braq AgBrs e cathode Agtaq e Ags AgsA...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write balanced half-reactions and devise a galvanic cell (write a cell diagram) to study each of the following reactions: (a) Pb(NO3)2(aq) + KSO4(aq) PbSO4(s) + 2 KNO3(aq), a precipitation reaction...
-
(a) Write balanced half-reactions for the redox reaction between sodium perchlorate and copper (I) nitrate in an acidic solution. (b) Write the balanced equation for the cell reaction and devise a...
-
(a) Write balanced half-reactions for the redox reaction of an acidified solution of potassium permanganate and iron(II) chloride. (b) Write the balanced equation for the cell reaction and devise a...
-
The following data are from the accounting records of Niles Castings for year 2: Units produced and sold . . . . . . . . . . . . . . . . . . . . . . . . . . . . 85,000 Total revenues and costs Sales...
-
EOQ, uncertainty, safety stock, reorder point, Clarkson Shoe Co. produces and sells excellent quality walking shoes. After production, the shoes are distributed to 20 warehouses around the country....
-
You are expected to have the gift bags in Problem ready at 5 P.M. However, you need to personalize the items (monogrammed pens, note pads, literature from the printer, etc.). The lead time is 1 hour...
-
I dont know you, and I very much liked working with Oscar Jones. Why was he transferred to Chicago? The customer you have just met for the first time is unhappy because of his relationship with the...
-
A process has the transfer function of Eq. 6-14 with K = 2, ?1 = 10, ?2 = 2. If ?a has the following value: Calculate the responses for a step input of magnitude 0.5 and plot them in a single figure....
-
Decision Trees at PVA For this TASK, you will work with a data set named PVA. It contains data that reflects charitable donations made to an American veterans' association. The data represents the...
-
Calculate the molar solubility of each substance in its respective solution: (a) Silver chloride in 0.20 m NaCl(aq); (b) Mercury(I) chloride in 0.150 m NaCl(aq); (c) Lead(II) chloride in 0.025 m CaCl...
-
The compound P 4 S 3 is oxidized by nitrate ions in acid solution to give phosphoric acid, sulfate ions, and nitric oxide, NO. Write the balanced equation for each half-reaction and the overall...
-
In a ventilation system, inside air at 34C and 70% relative humidity is blown through a channel, where it cools to 25C with a flow rate of 0.75 kg/s dry air. Find the dew point of the inside air, the...
-
he average monthly temperature (F) in Chicago, Illinois is shown in the table Month January February March April May June July August September October November December OF 25 28 36 48 61 72 74 75 66...
-
Find the intermediate points using DDA line algorithm. Assume the starting point (5, 6) and ending point (13, 10). b (6,7), (7,7), (9,9), (9,8), (11,9), (12,9), (12,10), (13, 10). O (6,7), (7,8),...
-
1. Figure 1 shows product structure for an assembly line. The value in percentages are the scrap rate for each process and and values in sec/unit are the processing time. The demand for the product...
-
WITH - Question 3: 12 x 12 24 marks) a) Determine the truth value of each of the following statements, and briefly jusily your answers. 1. VzeZ Bycz ((2) (x+y23) Truth value- 2. 3 nez VmeZ (EZ) Truth...
-
(10) In a BST with n nodes, what is the time complexity forsearching, insertion, and deletion in worst case, respectively?What is the time complexity for these operation in average case(balanced...
-
Many organizations implementing the balanced scorecard have a set of measures that capture the customer or external perspective. Sample measures in this category include customer satisfaction and...
-
What is the purpose of the journal wizard?
-
Starting with sodium azide as your source of nitrogen and using any other reagents of your choice, show how you would prepare each of the compounds in Problem 23.18. Compounds in 23.18 (a) (b) (c)...
-
Cinchocaine is a long-acting local anesthetic used in spinal anesthesia. Identify the most basic nitrogen atom in cinchocaine. N. N. Cinchocaine
-
For each pair of compounds, identify the stronger base. (a) (b) (c) vs. N. N. z. Vs. %3D N. N.
-
Explain the components of effective negative messages, including opening with a buffer, apologizing, showing empathy, presenting the reasons, cushioning the bad news, and closing pleasantly.
-
You have recently discovered a new planet orbiting a distant star in honor of your favorite physics professor you have decided to name the planet Drake based on observations you have determined that...
-
Which point is a solution to the following system of inequalities? (-2x+3y 1 -5+6ys1

Study smarter with the SolutionInn App


