Calculate the number of copper atoms in 2.45 mol of copper. SORT You are given the amount
Question:
Calculate the number of copper atoms in 2.45 mol of copper.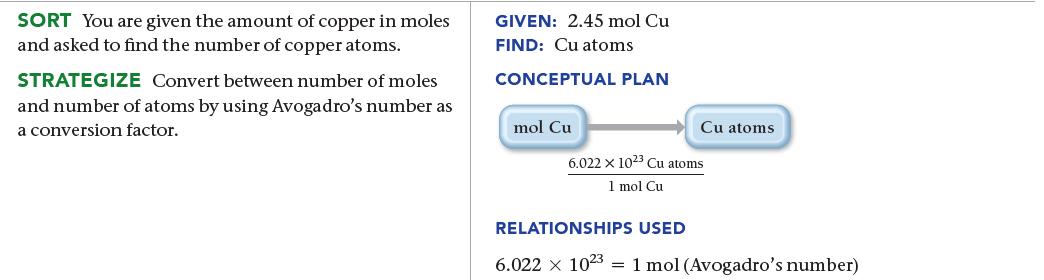
Transcribed Image Text:
SORT You are given the amount of copper in moles and asked to find the number of copper atoms. STRATEGIZE Convert between number of moles and number of atoms by using Avogadro's number as a conversion factor. GIVEN: 2.45 mol Cu FIND: Cu atoms CONCEPTUAL PLAN mol Cu Cu atoms 6.022 x 1023 Cu atoms 1 mol Cu RELATIONSHIPS USED 6.022 x 1023 1 mol (Avogadro's number)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
245 mol Cu x ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many copper atoms are in a copper penny with a mass of 3.10 g? (Assume that the penny is composed of pure copper.) SORT You are given the mass of copper and asked to find the number of copper...
-
In the experiment: Determining Avogadro's Number the values that I've got are Trial 1 Trial 2 Trial 2 Initial mass of Copper electrode (g) 12.517 12.480 12.434 Final mass of Copper electrode (g)...
-
(a) Suppose you have a cube of copper metal that is 0.236 cm on a side with a mass of 0.1206 g. If you know that each copper atom (radius = 128 pm) has a mass of 1.055 10 22 g (you will learn in...
-
At the beginning of the current season, the ledger of Highland Tennis Shop showed Cash $2,500; Inventory $1,700; and Common Stock $4,200. The following transactions were completed during April. Apr....
-
Are sunk costs ever differential costs?
-
Laurence Norton is the chief financial officer of Rotham Industries, a company that makes special-order sound systems for home theaters. His records for February revealed the following information:...
-
Environmental compliance: a market opportunity. Table 2.1 shows estimates of the size of the market for environmental technologies and services needed to comply with environmental standards. In 1996...
-
1. Discuss and contrast the features of the retirement plans offered by Analytical Solutions and Precision Manufacturing. 2. Which plan do you think is more desirable? Consider the features,...
-
A firm sells dinner table sets - each set includes one table and six chairs. The price of the set is $600. The variable cost of the table is $83, and that of each chair is $34. Fixed cost for the...
-
On May 1, Soriano Co. reported the following account balances along with their estimated fair values: On that day, Zambrano paid cash to acquire all of the assets and liabilities of Soriano, which...
-
What are the main ideas in Daltons atomic theory? How do they help explain the laws of conservation of mass, of constant composition, and of definite proportions?
-
An argon isotope has a mass number of 40 (A = 40). How many neutrons does it contain? (a) 40 (b) 18 (c) 22
-
Some lemon juice has a hydronium-ion concentration of 5.0 10-3 M. What is the pH of the lemon juice?
-
Define tolerable error.
-
Wil Stevens is executive vice president of a major automobile manufacturing company. Stevens was recently elected Mayor of Detroit. Prior to assuming office, he calls on you, his independent auditor,...
-
Identify three conditions that must exist before either difference or ratio estimation can be applied.
-
Jayne Phillips, CPA has been engaged by the town council of Granger to perform an audit of the town's June 30, 1999 financial statements in accordance with generally accepted auditing standards. By...
-
Under what condition is difference estimation more appropriate than ratio estimation?
-
During 2017, Away Company acquires a controlling interest in Stallward, Inc. Trial balances of the companies at December 31, 2017, are as follows: The following information is available regarding the...
-
Provide examples of a situations in which environmental disruptions affected consumer attitudes and buying behaviors.
-
Three of the compounds from Problem 13.14 can be prepared from the reaction between a hydride reducing agent (NaBH 4 or LAH) and a ketone or aldehyde. Identify those three compounds, and explain why...
-
For each of the following alkenes, assign the configuration of the double bond as either E or Z: a. b. c. d. F.
-
Draw the mechanism and predict the product of the following reaction. In this case, H 3 O + must be used as a proton source instead of water. Explain why. 1) xs MeMgBr 2) H*
-
In which of the following situations would it not be appropriate to use the following formula: PV= Co + C/(1+r) + C/(1+r)- +... +C/(1+r) when determining the present value (PV) of a cash flow stream?
-
KL Mobile is a major producer of automobiles in Kuala Lumpur. One of the divisions within KL Mobile is currently negotiating with another supplier regarding outsourcing component X that it...
-
what inventory amount appears on the firm's balance sheet as of March 31, 2022? Clothing retailer Forever Young has $1,000,000 of clothing in its warehouses as of December 31, 2021. Based on past...

Study smarter with the SolutionInn App


