Consider the reaction: Consider also this representation of an initial mixture of N 2 H 4 and
Question:
Consider the reaction:![]()
Consider also this representation of an initial mixture of N2H4 and N2O4: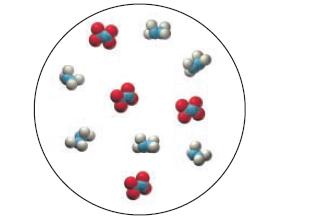
Which diagram best represents the reaction mixture after the reactants have reacted as completely as possible?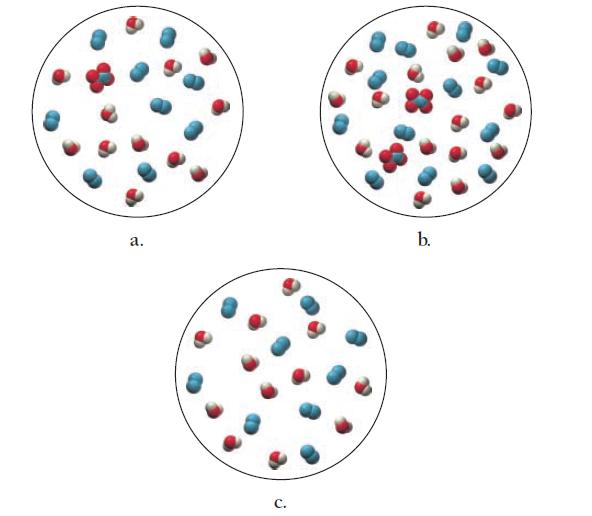
Transcribed Image Text:
2 N₂H4(8) + N₂O4(8) 3 N₂(g) + 4H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: A reaction mixture initially contains 5 moles of NO and 10 moles of H 2 . Without doing any calculations, determine which set of amounts best represents the mixture after the...
-
Nitrogen and hydrogen gas react to form ammonia according to the reaction: A flask contains a mixture of reactants represented by the image shown at the left. Which of the following images best...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Solve Prob. 410 using singularity functions. Use statics to determine the reactions. Prob. 410, The figure shows a cantilever consisting of steel angles size 100 Ã 100 Ã 12 mm mounted...
-
Riggins Trucking, Inc., has a current stock price of $41. For the past year, the company had net income of $5,150,000, total equity of $21,580,000, sales of $39,000,000, and 4.1 million shares of...
-
In a thin-slab, continuous casting process, molten steel leaves a mold with a thin solid shell, and the molten material solidifies as the slab is quenched by water jets en route to a section of...
-
The following scenarios describe situations faced by hypothetical companies that currently have a centralized organization structure. As you review each of the scenarios, provide your opinion as to...
-
Lopez Corporation incurred the following costs while manufacturing its product. Work in process inventory was $12,000 at January 1 and $15,500 at December 31. Finished goods inventory was $60,000 at...
-
Consider a web system for online shopping that will generate an ID for each order. The order ID is a sequence of 7 characters where each char- acter could be either one of the 26 upper case English...
-
Waymouth Manufacturing operates a contract manufacturing plant located in Dublin, Ireland. The plant provides a variety of electronics products and components to consumer goods manufacturers around...
-
Octane (C 8 H 18 ), a component of gasoline, reacts with oxygen to form carbon dioxide and water. Write the balanced chemical reaction for this process by passing a single piece of paper around your...
-
Hydrolysis of the compound B 5 H 9 forms boric acid, H 3 BO 3 . Fusion of boric acid with sodium oxide forms a borate salt, Na 2 B 4 O 7 . Without writing complete equations, find the mass (in grams)...
-
Derby Transportation Inc. designs and builds trains for rail networks across Canada. The company, which is publicly traded, has an October 31 year end. On March 15, 2020, Derby Transportation signed...
-
In relation to the below link regarding ''Going with the Flow: Agile Development at Dell" and using the major guidelines stated below please provide a written report on 1. what went wrong during the...
-
In recent years, many companies have been creating service-added competitive strategies to their product offerings. What is the main benefit of this approach? 1 It provides customers with access to...
-
Discuss the following topic(s) Research Title Introduction Literature Review Methodology Data Analysis
-
In the Men we carry in Our Minds by Sanders, what way of thinking has changed for him and why did it change? You can also use the resources in the Unit II Suggested Reading section to assist you with...
-
Please, discuss the following segmentation models: AIO, VALS, Sinus Milieus.
-
Phil Fritz is a financial consultant to Magna Properties Inc., a real estate syndicate. Magna Properties Inc. finances and develops commercial real estate (office buildings). The completed projects...
-
F.(3e* -2x 3 sin(2x)) is equal to 2 3 Cos 8. IT 3, t (4+@ 2 3, 1+o 1 4 Cos 4 4 1 3. 1 +4cos V7 (1+o 4 1 4 Cos 4 1+0 4-
-
Is the following statement true of false? If it is false, rephrase it so that it is true. The entropy of a system cannot increase in an adiabatic process.
-
Which of the following processes is spontaneous? a. The reversible isothermal expansion of an ideal gas. b. The vaporization of superheated water at 102C and 1 bar. c. The constant pressure melting...
-
One joule of work is done on a system, raising its temperature by one degree centigrade. Can this increase in temperature be harnessed to do one joule of work? Explain.
-
Statement of cash flows-indirect method The comparative balance sheet of Merrick Equipment Co. for December 31, 20Y9 and 20Y8, is as follows: December 31, 20Y9 December 31, 20Y8 Assets Cash Accounts...
-
Thunderduck Shoes provides shoe shining and repair services to customers. For the year which ended Dec 31, the company reports the following amounts: Account Amount Account Amount Rent Expense 22,400...
-
Shelby Woods owns and operates an ice cream factory, the finest of its kind in all the world. Given that pints of ice cream are meant to be identical (for the same flavor), her company uses process...

Study smarter with the SolutionInn App


