Nitrogen and hydrogen gas react to form ammonia according to the reaction: A flask contains a mixture
Question:
Nitrogen and hydrogen gas react to form ammonia according to the reaction:![]()
A flask contains a mixture of reactants represented by the image shown at the left.
Which of the following images best represents the mixture in the flask after the reactants have reacted as completely as possible? What is the limiting reactant? Which reactant is in excess?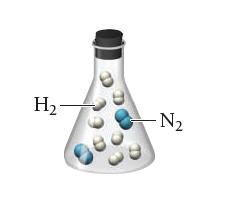
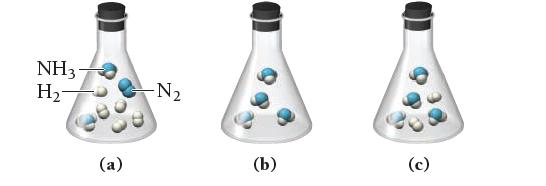
Transcribed Image Text:
N₂(g) + 3 H₂(g) →→→ 2 NH3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
c Nitrogen is the limiting reactant and t...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Nitrogen (N2) and hydrogen (H2) react to form ammonia (NH3). Consider the mixture of N2 and H2 shown in the accompanying diagram. The blue spheres represent N, and the white ones represent H. Draw a...
-
Nitrogen and hydrogen gases react to form ammonia gas as follows: N2 (g) + 3 H2 2NH3 (g) At a certain temperature and pressure, 1.2 L of N2 reacts with 3.6 L of H2. If all the N2 and H2 are...
-
Ammonia is created in the Haber process in a rigid container (nitrogen gas plus hydrogen gas react to form ammonia gas) at a constant temperature. 5 moles of hydrogen gas are mixed with 10 moles of...
-
We consider the following CFG SE + SIE E 01|2|3|4|5|67|8|9|(S) Apply the leftmost derivation and rightmost derivation with a top-down parser for this sentence: (3+7+ (1+4)) + 2
-
The financial statements of Hershey Foods are presented in Appendix B, following the financial statements for Tootsie Roll Industries in Appendix A. Instructions (a) Based on the information in these...
-
A sphere of 25-mm diameter contains an embedded electrical heater. Calculate the power required to maintain the surface temperature at 94C when the sphere is exposed to a quiescent medium at 20C for:...
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
Callie Peters is completing the audit of MakingNewFriends.com for the year ended December 31, 2013. Callie has been the audit manager on this engagement for the past three years. MakingNewFriends.com...
-
QUESTION IO.17 (Applicationofdecline invaluemethods) George Bensonisa45-year-oldresident taxpayerwhohasconducted businessasanITProgrammerfor8yearsandhasannualturnover below...
-
John Campbell, an employee of Manhattan Construction Company, claims to have injured his back as a result of a fall while repairing the roof at one of the Eastview apartment buildings. He filed a...
-
What is reaction stoichiometry? What is the significance of the coefficients in a balanced chemical equation?
-
Ammonia, NH 3 , can be synthesized by the reaction: Starting with 86.3 g NO and 25.6 g H 2 , find the theoretical yield of ammonia in grams. 2 NO(g) + 5 H(g) 2 NH3(g) + 2 HO(g)
-
Send a letter to Grandma and convince her that whatever electric shocks she may have received over the years have been due to the movement of electrons already in her body-not electrons from...
-
A company receives a $900 utility bill for the current month but does not plan to pay the bill until early next month. Record the receipt of the utility billusing (a) accrual-basis accounting and (b)...
-
Explain why workload management is important for safe, effective, and efficient delivery of health care.
-
Define two cost accounting methods. Describe typical uses for each and include examples. Discuss which of the various cost accounting methods you think are most appropriate for measuring the costs of...
-
Toys For You is considering a project for its capital budget. While the project's NPV is estimated at $650,000, this estimate assumes that the economy and market conditions will be average over the...
-
How was the Trump/Clinton election in 2016 impacted by voter laws and the media?
-
Merchandise is sold on account to a customer for $12,500, terms FOB shipping point, 1/10, n/30. The seller paid the freight of $250. Determine the following: (a) Amount of the sale, (b) Amount...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
The observed Boyle temperatures of H 2 , N 2 , and CH4 are 110, 327, and 510. K, respectively. Compare these values with those calculated for a van der Waals gas with the appropriate parameters.
-
For the Berthelot equation, V m = (RT/P) + b a (RT 2 ) find an expression for the Boyle temperature in terms of a, b, and R.
-
For a van der Waals gas, z = V m /(V m b) a/RTV m . Expand the first term of this expression in a Taylor series in the limit V m >> b to obtain z 1 + (b a/RT)(1/V m ).
-
Why is the relational model more important above the other models?
-
Discuss why people often resist new ideas such as the Relational Model in Business Logic ?
-
Who developed the relational model, when, and why?

Study smarter with the SolutionInn App


