Consider this overall reaction, which is experimentally observed to be second order in AB and zero order
Question:
Consider this overall reaction, which is experimentally observed to be second order in AB and zero order in C:![]()
Is the following mechanism valid for this reaction?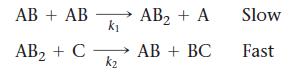
Transcribed Image Text:
AB + C A + BC
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider this overall reaction, which is experimentally observed to be second order in X and first order in Y: a. Does the reaction occur in a single step in which X and Y collide? b. Is this...
-
The reaction AB(aq) A( g) + B( g) is second order in AB and has a rate constant of 0.0118 M -1 s -1 at 25.0 C. A reaction vessel initially contains 250.0 mL of 0.100 M AB that is allowed to react...
-
Consider the following reaction: The following rate equation has been experimentally established for this process: Rate = k [HO - ] [CH 3 CH 2 Br] The energy diagram for this process is shown below:...
-
What (if anything) is wrong with each of the following statements? a. if (a > b) then c = 0; b. if a > b { c = 0; } c. if (a > b) c = 0; d. if (a > b) c = 0 else b = 0;
-
An investment banker is analyzing two companies that specialize in the production and sale of candied yams. Traditional Yams uses a labor-intensive approach, and Auto-Yams uses a mechanized system....
-
Wilco Corporation has the following account balances at December 31, 2012. Share capitalordinary, $5 par value ....$ 510,000 Treasury shares ..............90,000 Retained earnings...
-
Political expectations, inflation, and unemployment Consider a country with two political parties, Democrats and Republicans. Democrats care more about unemployment than Republicans, and Republicans...
-
The ledger of Costello Company at the end of the current year shows Accounts Receivable $110,000, Sales Revenue $840,000, and Sales Returns and Allowances $20,000. Instructions (a) If Costello uses...
-
As part of a survey, 15 adults were asked, "How many hours did you spend at your job last week?" The results are shown in the stem-and-leaf display below. Use the display to answer the questions that...
-
If a temperature increase from 20.0 C to 35.0 C triples the rate constant for a reaction, what is the value of the activation barrier for the reaction?
-
If a temperature increase from 10.0 C to 20.0 C doubles the rate constant for a reaction, what is the value of the activation barrier for the reaction?
-
Data Set 15 Passive and Active Smoke in Appendix B includes measured serum cotinine levels (mg/mL) from the three groups of subjects: 902 smokers, 433 nonsmokers exposed to tobacco smoke, and 358...
-
What potential problems will a marketer encounter in having a global advertising campaign?
-
General Motors (GM) is a global US car company which produced 9.8 million vehicles in 2015, sold in 125 countries, split between ten brands: Chevrolet, Buick, GMC, Cadillac, Opel, Vauxhall, Holden,...
-
What are the consequences for the global economy of the global economic/political power of China?
-
What are the benefits of having a geographical organization?
-
Founded in 1995 by Mr Wang Chuanfu in Shenzhen, Southern China, BYD (Build Your Dreams) is today the largest supplier of rechargeable batteries in the world and has diversified into electronic...
-
Ebanks Company has a line of credit with United Bank. Ebanks can borrow up to $200,000 at any time over the course of the 2013 calendar year. The following table shows the prime rate expressed as an...
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
When the input to a system is a unit step function, the response is 120 cos 2tu(t). Obtain the transfer function of the system.
-
For the RLC circuit shown in Fig. 16.88 , find the complete response if v(0) = 100 V when the switch is closed. t = 0 1H 6 w 100 cos 4t V (+ -
-
The switch in Fig. 16.87 moves from position 1 to position 2 at t = 0. Find v(t), for all t > 0. t= 0 2, 15 V (+ 10 mF 0.25 H +1)
-
Seven and a half years ago, Chris invested $10,000 in a retirement fund that grew at the rate of 6.82%/year compounded quarterly. What is his account worth today?
-
A College Alcohol Study interviewed an SRS of 1000 college students about their drinking habits. Suppose that half of all college students "drink to get drunk" at least once in a while. That is...
-
For a bivariate analysis involving the two variables X and Y, if (xx)(y) = -91, n = 42, 8 = 1.47 and Sy = 2.72, calculate the covariance, Cov(X,Y). X

Study smarter with the SolutionInn App


