Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and
Question:
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent.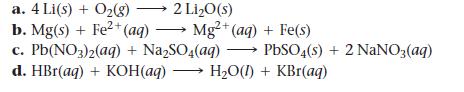
Transcribed Image Text:
a. 4 Li(s) + O₂(g) → 2 Li₂O(s) b. Mg(s) + Fe²+ (aq)→ Mg2+(aq) + Fe(s) c.Pb(NO3)2(aq) + Na₂SO4(aq) d. HBr(aq) + KOH(aq)→ H₂O(1) + KBr(aq) PbSO4(s) + 2NaNO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Redox reaction oxidizing agen...View the full answer

Answered By

Ajeet Singh
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
4+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Consider the redox reaction (a) Identify the oxidizing agent on the left side of the reaction and write a balanced oxidation half-reaction. (b) Identify the reducing agent of the left side of the...
-
stock options are no longer as valuable and employees in startups are losing out to founders and early investors for all of these reasons except: there is higher employee turnover, less of a delay in...
-
1. Consider a household that has $300 to spend on back-to- school shoes. They visit one of the discount shoe stores where shoes are buy one pair for $50, get the second half off. a. Draw the budget...
-
The Palmer Company manufactures various types of clothing products for women. To accumulate the costs of manufacturing these products, the companys accountants have established a computerized cost...
-
Determine the reactions at the supports A, C, and E of the compound beam. 3 kN/m 12 KN
-
What is meant by corporate governance and why is it important?
-
On January 3, 2012, Trusty Delivery Service purchased a truck at a cost of $90,000. Before placing the truck in service, Trusty spent $3,000 painting it, $1,500 replacing tires, and $4,500...
-
1. A car has tires that have an outer diameter of 31 inches. If the wheels are turning with an angular velocity of 12 rad/s, how far in miles will the car travel in 2 hours? Enter your result rounded...
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b. Ni(s) + Pb+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn+ (aq) 2+ Ni+(aq) + Zn(s) Ni+(aq) +...
-
What is the oxidation state of Cl in each ion? a. CIO- b. ClO c. ClO3 d. CIO4
-
A travel agency will plan a tour for groups of size 25 or larger. If the group contains exactly 25 people, the cost is $500 per person. However, each person's cost is reduced by $10 for each...
-
What are the principal inputs and outputs of the HR management process as reflected in the systems flowchart in Figure 14.4 (pg. 531)? FIGURE 14.4 Human Resources Management Process Systems Flowchart...
-
Having decided to use his most likely scenario as the plan for 20x7, Jack decides to develop cash forecasts. The opening balance sheet is presented below. OTHER INFORMATION: The company pays its...
-
From the following balance sheet pass opening entry in the beginning of next accounting year. Liabilities Owner's Capital Secured Bank Loan Bank Overdraft Sundry Creditors Tax payable Outstanding...
-
Refer to the table for Beverly Entertainment Enterprises in part (1) of Problem 3-9. Required 1. Prepare a trial balance at October 31. 2. Prepare an income statement for the month of October. 3....
-
At what temperature does water boil on the top of Mount Everest, elevation z = 8848 m? Recall that the dependence of pressure with altitude is given by: where, Patm is atmospheric pressure, g is the...
-
On December 1 of the current year, Jordan, Inc., assigns $125,000 of its accounts receivable to McLaughlin Company for cash. McLaughlin Company charges a $750 service fee, advances 85% of Jordans...
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
In a falling-ball viscometer, a steel ball 1.6 mm in diameter is allowed to fall freely in a heavy fuel oil having a specific gravity of 0.94. Steel weighs 77 kN/m 3 . If the ball is observed to fall...
-
A capillary tube viscometer similar to that shown in Fig. 2.7 is being used to measure the viscosity of an oil having a specific gravity of 0.90. The following data apply: Tube inside diameter = 2.5...
-
In a falling-ball viscometer, a steel ball with a diameter of 0.063 in is allowed to fall freely in a heavy fuel oil having a specific gravity of 0.94. Steel weighs 0.283 lb/in 3 . If the ball is...
-
Can anyone in the class explain what a sale/leaseback arrangement is andwhy a managerial accountant would be interested in such an arrangement? What would be the advantages and disadvantages of this...
-
What are the recipients of the work expected to abide by, according to the copyright notice?
-
Compare and contrast long-form and short-form reports. Consider the following in your response: Explain when each report type should be used. Explain the purpose of the components that should be...

Study smarter with the SolutionInn App


