Pick an appropriate solvent from Table 14.3 to dissolve each substance. State the kind of intermolecular forces
Question:
Pick an appropriate solvent from Table 14.3 to dissolve each substance. State the kind of intermolecular forces that would occur between the solute and solvent in each case.
a. Isopropyl alcohol (polar, contains an OH group)
b. Sodium chloride (ionic)
c. Vegetable oil (nonpolar)
d. Sodium nitrate (ionic)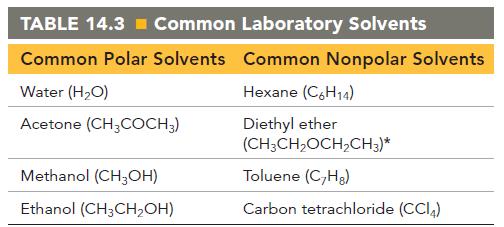
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





