Rank each set of substances in order of increasing standard molar entropy (S). Explain your reasoning. a.
Question:
Rank each set of substances in order of increasing standard molar entropy (S°). Explain your reasoning.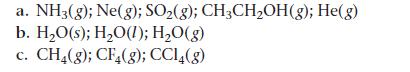
Transcribed Image Text:
a. NH3(g); Ne(g); SO₂(g); CH3CH₂OH(g); He(g) b. H₂O(s); H₂O(1); H₂O(g) C. CH4(g); CF4(g); CC14(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a He Ne SO 2 NH 3 CH 3 CH 2 OH From He to Ne there is an ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank each set of substances in order of increasing standard molar entropy (S). Explain your reasoning. a. 1(g); F(g); Br2(g); Cl(g) b. HO(g); HO(g); HS(g) c. C(s, graphite); C(s, diamond); C(s,...
-
Rank each set of compounds in order of increasing acidity: a. b.
-
Rank each set of compounds in order of increasing boiling points. (a) Triethylamine, di-n-propylamine, n-propyl ether (b) Ethanol, dimethylamine, dimethyl ether (c) Diethylamine, diisopropylamine,...
-
What are the costs of healthcare, where does the money come from, and where is it spent?
-
Discuss the economic and trade importance of the big emerging markets.
-
The Safety Alarm Corporation manufactures and sells a patented electronic device for detecting burglaries. The firm uses return on investment as a measure for the control of operations for each of...
-
Petitioner Christy Brzonkala met respondents Antonio Morrison and James Crawford at a campus party at Virginia Polytechnic Institute (Virginia Tech), where they were all students. At the party, the...
-
Ratio Computations and Effect of Transactions Presented below is information related to Leland Inc. (a) Compute the following ratios or relationships of Leland Inc. Assume that the ending account...
-
Using the list method or the switch method, find the inverse function of: f(x)=6x+1-13 f1(x) =
-
Use data from Appendix IIB to calculate S rxn for each of the reactions. In each case, try to rationalize the sign of S rxn . Appendix IIB a. CH4(g) + H(g) CH6(g) b. C(s) + HO(g) CO(g) + H(g) c....
-
What is the molar entropy of a pure crystal at 0 K? What is the significance of the answer to this question?
-
The Coca-Cola Company pulled a UK Internet promotion campaign after parents accused it of targeting children by using references to a notorious pornographic movie. As part of its efforts to reach...
-
1. What is leadership, and why is leadership vital to successful health organizations? Write a paragraph that supports your definition and another paragraph explaining why health organizations need...
-
Conduct an analysis of the communication strategy of General Motors and its leadership. What are the strengths and weaknesses observed in this strategy?
-
How are you being influenced and persuaded? In what ways are you being influenced? 0 Who or what influences you to take action? 0 Are you always aware of when you are being influenced? 0 What are...
-
a. As a business student, why is it important to know the process of corporate re-engineering and re-structuring strategies in conducting business? b. Why do corporation expand their operations in...
-
What are the core principles in the (ECA Code of Ethics) that reflect a commitment to working collaboratively with families and children to build respectful relationships.
-
Describe and discuss the five major suggestions for using other people's criticisms of us as a means of personal growth.
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
Construct a graph of the angular velocity of a car wheel as a function of time. (a) Assume the wheel starts from rest and moves with a constant (center of mass) velocity. (b) Assume the car starts...
-
Two crates of mass 5.0 kg and 9.0 kg are connected by a rope that runs over a pulley of mass 4.0 kg as shown in Figure P8.57.? (a) Make a sketch showing all the forces on both crates and the pulley.?...
-
Two crates of mass m 1 = 15 kg and m 2 = 25 kg are connected by a cable that is strung over a pulley of mass m pulley = 20 kg as shown in Figure P8.58. There is no friction between crate 1 and the...
-
I have built a proportional control system to replace the "resume speed" function in the cruise control on my Tesla. It applies an acceleration that is proportional to the difference between the...
-
The internal forces within the train (the forces each car exerts on its neighbors) are equal and opposite, according to Newton's third law. This means they cancel each other out when considering the...
-
A student leaving campus for spring break wants to make sure the air in his dorm room has a high water vapor pressure so that his plants are comfortable. The dorm room measures 3.45 m 4.64 m 3.63 m...

Study smarter with the SolutionInn App


