The phase diagram for sulfur is shown here. The rhombic and monoclinic states are two solid states
Question:
The phase diagram for sulfur is shown here. The rhombic and monoclinic states are two solid states with different structures.
a. Below what pressure does solid sulfur sublime?
b. Which of the two solid states of sulfur is more dense?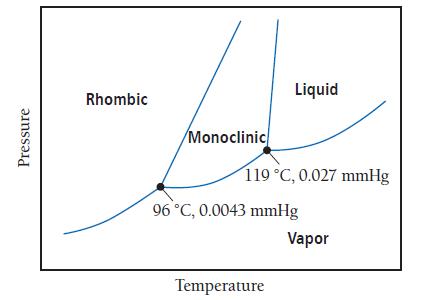
Transcribed Image Text:
Pressure Rhombic Monoclinic Liquid 119 °C, 0.027 mmHg 96 °C, 0.0043 mmHg Temperature Vapor
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 00...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the accompanying phase diagram for sulfur to answer the following questions. (The phase diagram is not to scale.) a. How many triple points are in the phase diagram? b. What phases are in...
-
Why are there no points in the phase diagram for sulfur in Figure 8.11 that show rhombic and monoclinic solid phases in equilibrium with liquid and gaseous sulfur? Figure 8.11 Liquid Monoclinic-...
-
Look at the phase diagram for sulfur in Figure 11.12, bottom. What states of sulfur would you expect to see at each of the triple points? What solid phase would you expect to freeze out if you cooled...
-
The combined sewer system in city ABC is comprised of two parallel interceptors referred to as "North" and "South" lines. The southern line is connected to a newly built wastewater treatment plant....
-
The Federal Crop Insurance Corporation (FCIC) was created as a wholly government-owned corporation to insure wheat producers against unavoidable crop failure. As required by law, the FCIC published...
-
Design an op-amp circuit that has the following input/output relationship: Vo = 5V1 + 0.5V2.
-
What does argv provide to our program?
-
Prepare a flowchart of the field service division process at DEE as described here. Start from the point where a call is received and end when a technician finishes the job. DEF was a multibillion...
-
Even method: 1- Algorithm Compare between recursion and iteration 2- Memory representation (Ex: EVEV (0,10)) recursion iteration
-
The high-pressure phase diagram of ice is shown here. Notice that, under high pressure, ice can exist in several different solid forms. What three forms of ice are present at the triple point marked...
-
Show how the fluorite structure accommodates a cation-toanion ratio of 1:2.
-
The wave functions of Problem 58, as well as their derivatives, need to be continuous at x = L if these functions are to represent the quantum state of a particle in the finite square well.(a) Show...
-
pick answer for example 1- T1, T2 and example 2-T1, T2 and there is a option to pick from. thank you/ Below are two examples of concurrent transactions. Select the type of conflict, if any, executed...
-
A piezoelectric bimorph operating as a bender actuator has the cross-sectional geometry shown in Figure 4.42. (a) Compute the nondimensional curvature as shown in equation (4.134) for this geometry....
-
If total liabilities decreased by $30400 and owner's equity increased by $21000 during a period of time, then total assets must change by what amount and direction during that same period?
-
Permata Sdn Bhd manufactures and sells its single Product X through manufacturer agents. The agents are paid a commission of 20% of the selling price. In the coming financial year, the sales agents'...
-
Pound has $ 44 comma 000 capital and Flores has $ 26 comma 000 capital in the Pound & Flores partnership. Pound and Flores share profits and losses equally. Ritter Reese contributes cash of $ 36...
-
IPort Products makes cases for portable music players in two processes, cutting and sewing. The cutting process has a capacity of 150,000 units per year; sewing has a capacity of 180,000 units per...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
It is believed that penicillin antibiotics are biosynthesized from amino acid precursors. Identify the two amino acids that are most likely utilized during the biosynthesis of penicillin antibiotics:...
-
Draw the s-cis conformation of the dipeptide Phe-Phe and identify the source of steric hindrance.
-
Using a bond-line structure, show the tetrapeptide obtained when two molecules of Cys-Phe are joined by a disulfide bridge.
-
Using the data in the chart below, what is the expected return if you have 50% of the portfolio in Stock A and 50% of the portfolio in Stock B? Stock Expected Return Stock A 15% Stock B 10%...
-
A 20-Kilogram car traveling east at 6 meters per second collides with a 30-Kilogram car that is traveling west. If both the cars come to rest immediately after the collision, what was the speed of...
-
How much energy is required to melt 929 g of ice into liquid water at 0 C? The heat of fusion for water is 333.6 J/g. energy required: TOOLS x10 J

Study smarter with the SolutionInn App


