This reaction has an equilibrium constant of K p = 2.26 * 10 4 at 298 K.
Question:
This reaction has an equilibrium constant of Kp = 2.26 * 104 at 298 K.![]()
Calculate Kp for each reaction and predict whether reactants or products will be favored at equilibrium.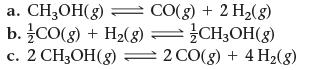
Transcribed Image Text:
CO(g) + 2 H₂(g) = CH3OH(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a 442 x 105 react...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
This reaction has an equilibrium constant of Kp = 2.2 * 10 6 at 298 K. Calculate Kp for each reaction and predict whether reactants or products will be favored at equilibrium. 2 COF2(g) = CO(g) +...
-
A particular reaction has an equilibrium constant of K p = 0.50. A reaction mixture is prepared in which all the reactants and products are in their standard states. In which direction does the...
-
The following reaction has an equilibrium constant Kc equal to 3.07 104 at 24oC. For each of the following compositions, decide whether the reaction mixture is at equilibrium. If it is not, decide...
-
The RRR Company has a target current ratio of 2.4. Presently, the current ratio is 3.3 based on current assets of $6,567,000. If RRR expands its inventory using short- term liabilities (maturities...
-
Explain the important factors to consider for capital investment decisions relating to advanced technology and P2 opportunities.
-
How will you respond to Tim's question?
-
Helen Cole, her husband, and her son were partners in a business. Mrs. Cole's husband died, leaving his equity in the partnership to Mrs. Cole. Mrs. Cole and her son plan to form a new partnership...
-
Essence of Persia, Inc., began operations on January 1, 2010. The company produces a hand and body lotion in an eight-ounce bottle called Eternal Beauty. The lotion is sold wholesale in 12-bottle...
-
Consider the following information: 'a company losing two ships during a storm'. This is an example of a/an: internal transaction. new condition. external transaction. underlying condition. event
-
A chemist trying to synthesize a particular compound attempts two different synthesis reactions. The equilibrium constants for the two reactions are 23.3 and 2.2 * 10 4 at room temperature. However,...
-
Ethene (C 2 H 4 ) can be halogenated by this reaction: where X 2 can be Cl 2 (green), Br 2 (brown), or I 2 (purple). Examine the three figures representing equilibrium concentrations in this reaction...
-
Major League Baseball rules require that the balls used in baseball games must have circumferences between 9 and 9.25 inches. Suppose the balls produced by the factory that supplies balls to Major...
-
1. What are the differences between Directional, Market Neutral, and Event-Driven Strategies? 2. What are the differences between Absolute return and relative return measurements?
-
The Millard Division's operating data for the past two years are provided below: Return on investment Net operating income Turnover Margin Sales Year 1 Year 2 12% 36% ? $ 540,000 3 ? ? $ 3,290,000 ?...
-
[15] For the curve defined by the following pair of parametric equations. x(t)= cos( t), y(t)=sin(2x 1), 0sts 2 a. Identify the point on the graph for which the y-value is a maximum. What is the...
-
Crane produces two joint products, Coconut Milk and Coconut Meat. The company uses the leftover Coconut Skin as a by-product. All three are produced from joint costs of $81500: direct materials...
-
"Financial advisor Suze Orman says one thing about housing while Warren Buffett (the Oracle of Omaha) says another. Which one do you agree with ... AND WHY??
-
For the decision of Exercise 11, youve just learned that you are on the short list and now estimate the chance that youll be called for an interview is 0.70. Does that change your choice of actions?...
-
Write a while loop that uses an explicit iterator to accomplish the same thing as Exercise 7.3. Exercise 7.3. Write a for-each loop that calls the addInterest method on each BankAccount object in a...
-
Evaluate R eq looking into each set of terminals for each of the circuits shown in Fig. 2.103 . a. b. 62 ww- 3 k2 2 k2 6 k2 ww 6 k2
-
Find i and V o in the circuit of Fig. 2.100 . 80 2 24 Q ww 25 2 30 : 20 20 V V. 20 2 60
-
Calculate V o and I o in the circuit of Fig. 2.99. 30 70 200 V 20 5
-
A precision lathe costs $20,000 and will cost $30,000 a year to operate and maintain. If the discount rate is 12% and the lathe will last for 5 years, what is the equivalent annual cost of the tool?...
-
Use the following information to complete Schedule M-1 (Form 1120) for ADZ Corporation. ADZ net income per books $350,485 Federal income taxes 98,389 Excess of capital losses over capital gains 3,867...
-
A friend at university notes that you are studying Finance and Investment Management. He asks why ( FNB ) the First National Bank was trading at a share price of R 4 7 . 3 3 and ( STD ) Standard Bank...

Study smarter with the SolutionInn App


