Use the BornHaber cycle and Table 10.3 to calculate the lattice energy of CaO. (H sub for
Question:
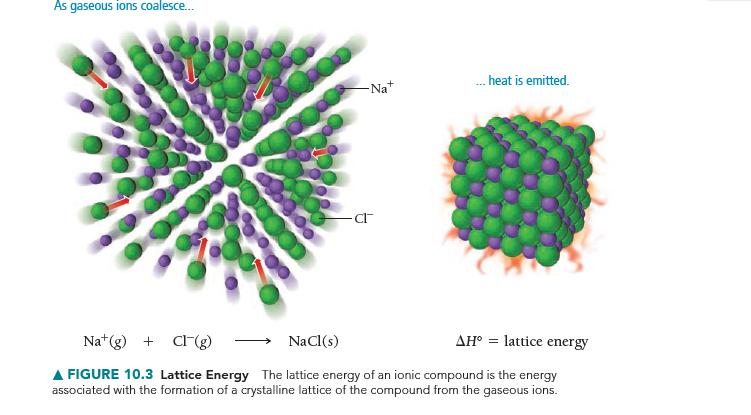
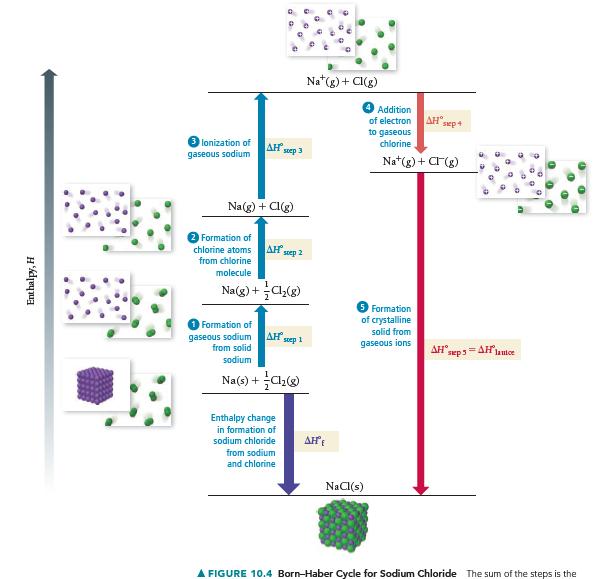
Use the Born–Haber cycle and Table 10.3 to calculate the lattice energy of CaO. (ΔHsub for calcium is 178 kJ/mol; IE1 and IE2 for calcium are 590 kJ/mol and 1145 kJ/mol, respectively; EA1 and EA2 for O are -141 kJ/mol and 744 kJ/mol, respectively.)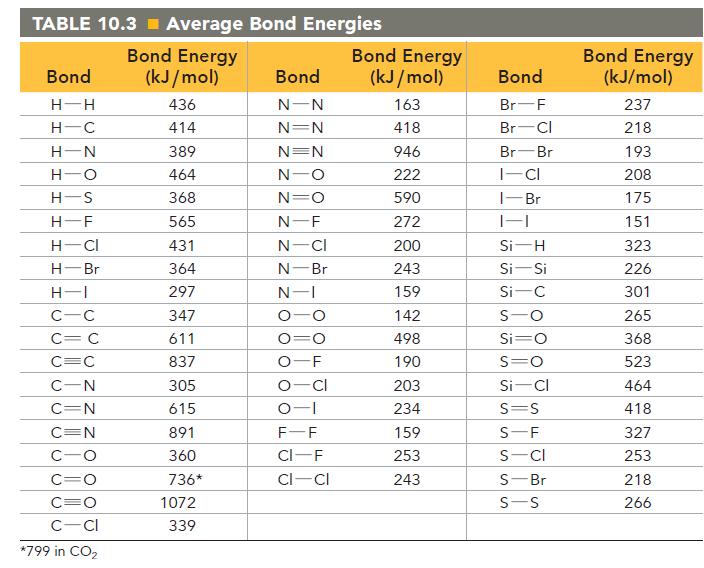


Transcribed Image Text:
TABLE 10.3 Average Bond Energies Bond Energy (kJ/mol) 436 414 389 464 368 565 431 364 297 347 611 837 305 615 Bond H-H H-C H-N H-O H-S H-F H-CI H-Br H-1 C-C C = C C=C C-N C=N C=N C-O C=O C=O C-CI *799 in CO₂ 891 360 736* 1072 339 Bond N-N N=N N=N N-O N=O N-F N-CI N-Br N-I O -O CI -1 O F-F CI-F CI-CI Bond Energy (kJ/mol) 163 418 946 222 590 272 200 243 159 142 498 190 203 234 159 253 243 Bond Br-F Br Cl Br Br I-CI 1-Br 1-1 Si-H Si-Si Si-C S-O Si=O S=O Si-Cl S=S S-F S-CI S-Br S-S Bond Energy (kJ/mol) 237 218 193 208 175 151 323 226 301 265 368 523 464 418 327 253 218 266
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The BornHaber cycle relates the lattice energy of an ionic compoun...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
"Hello to everyone. I try to calculate the lattice energy of d-fructose using habit 98 and use different radius. i expect to get difference lattice energy and electrostatic interaction for each...
-
From the ionic radii given in Figure 7.7, calculate the potential energy of a Ca2+ and O2- ion pair that is just touching (the magnitude of the electronic charge is given on the back inside cover)....
-
Energy is required to remove two electrons from Ca to form Ca 2+ , and energy is required to add two electrons to O to form O 2 - . Yet CaO is stable relative to the free elements. Which statement is...
-
Suppose that a 20-year bond pays $4 coupon once a year, and its yield rate is 6%. The bond has write-down of $0.49 for the first year. Calculate the write-down amount for the third year. Round the...
-
What is an entity-relationship diagram? Describe some symbols used in ER modeling, and explain the function of each one.
-
Given a risk-free rate (rËf) of 6 percent and a market risk premium (rËm rËf) of 8.5 percent, calculate the required rate of return on each of the following stocks, based on the betas...
-
What are the main characteristics of the conventional accounting and ecological accounting categories? Why is the distinction necessary?
-
In 2011, Alliant Corporation acquired Centerpoint Inc. for $300 million, of which $50 million was allocated to goodwill . At the end of 2013, management has provided the following information for a...
-
From this link: https://www.zawya.com/en/press-release/events-and-conferences/the-worlds-largest-esports-festival-is-coming-back-this-summer-with-more-than-double-the-prize-pool-hzqy4wv4 Read the...
-
Explain the difference between endothermic reactions and exothermic reactions with respect to the bond energies of the bonds broken and formed.
-
What is bond energy? How can you use average bond energies to calculate enthalpies of reaction?
-
Factor each polynomial. If a polynomial cannot be factored, write prime. Factor out the greatest common factor as necessary. 9p 2 24p + 16
-
A spring with a spring constant k = 3 . 7 N / m ( note the units ) is hung vertically from a hook. A weight is hung on the other end of the spring, and the spring stretches by 1 . 2 cm ( note the...
-
2. Consider a portfolio that includes 10 private loans of $120,000 each and 200 bonds that costs $1500 each. Each private loan has a duration of 4 and convexity of 8.5 and each bond have a duration...
-
Look up the FBI UCR data for the most recent complete year of data, which can be found at https://ucr.fbi.gov/crime-in-the-u.s/ Gather the data for Part I crimes in the city and state you live in as...
-
A 550 g basketball in flight has a velocity of 4.18 m/s [W] at the top of its arc. At that point it is struck by a 58.4 g arrow flying north at 38.2 m/s. The arrow becomes lodged in the ball. Based...
-
Plz help me answer these question. It related to Branding and Advertising. 1/ Advertising appeals A/ Describe the differences in the three main types of advertising messages - emotional (affective),...
-
Koontz, Ltd. had the following income statement for 2009 Required: (a) Prepare a break-even graph for Koontz, Ltd. (b) Prepare a proï¬t-volume graph for Koontz, Ltd. (c) Prepare a short...
-
You are a Loan Officer with an Investment Bank. Today you need to set your lending parameters. They are: LTV: 55% 10 Year T-Bill: TBD Rate Markup: 300 Basis Points Term: 30 Years Amortization: 30...
-
Use Fig. 4.47. The surface is 2.00 m long. Water 1.85 m 0.75-m radius
-
Use Fig. 4.48. The surface is 2.50 m long. Ammonia sg = 0.826 0,62 m 1.25 m
-
Use Fig. 4.49. The surface is 5.00 ft long. Water 10.00 ft 75 15.00 ft
-
Required information [The following information applies to the questions displayed below.] Bunnell Corporation is a manufacturer that uses job-order costing. On January 1, the company's inventory...
-
Focuses on Organizational Behavior and the application of theory to the workplace. Take two concepts or theories covered in this course and apply the concepts or theories to your current or past...
-
Unit sales for new product ABC have varied in the first seven months of this year as follows: Month Unit Sales Jan Feb Mar Apr May Jun Jul 248 482 341 174 456 310 273 What is the (population)...

Study smarter with the SolutionInn App


