Use the data in Appendix II, Table B to calculate H for the formation of a 1
Question:
Use the data in Appendix II, Table B to calculate ΔH° for the formation of a 1 M solution of H2SO4 from SO3(g).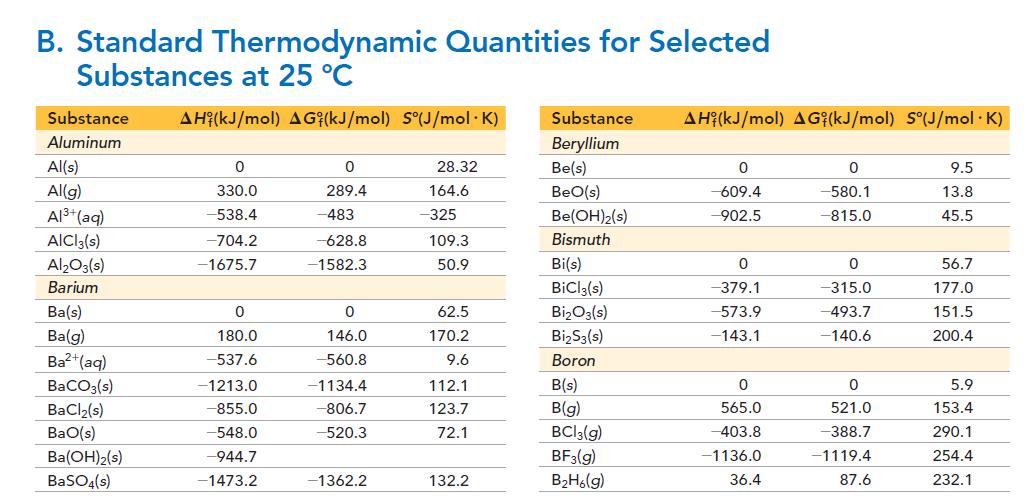
Transcribed Image Text:
B. Standard Thermodynamic Quantities for Selected Substances at 25 °C Substance AH (kJ/mol) AG (kJ/mol) S°(J/mol.K) Aluminum Al(s) Al(g) Al³+ (aq) AICI 3(s) Al₂O3(s) Barium Ba(s) Ba(g) Ba²+ (aq) BaCO3(s) BaCl₂(s) BaO(s) Ba(OH)2(s) BaSO4(s) 0 330.0 -538.4 -704.2 -1675.7 0 180.0 -537.6 -1213.0 -855.0 -548.0 -944.7 -1473.2 0 289.4 -483 -628.8 -1582.3 0 146.0 -560.8 -1134.4 -806.7 -520.3 -1362.2 28.32 164.6 -325 109.3 50.9 62.5 170.2 9.6 112.1 123.7 72.1 132.2 Substance AH (kJ/mol) AGi(kJ/mol) S°(J/mol .K) Beryllium Be(s) BeO(s) Be(OH)2(s) Bismuth Bi(s) BiCl3(s) Bi₂O3(s) Bi₂S3(s) Boron B(s) B(g) BC13(g) BF3(g) B₂H6(g) 0 -609.4 -902.5 0 -379.1 -573.9 -143.1 0 565.0 -403.8 -1136.0 36.4 0 -580.1 -815.0 0 -315.0 -493.7 -140.6 0 521.0 -388.7 -1119.4 87.6 9.5 13.8 45.5 56.7 177.0 151.5 200.4 5.9 153.4 290.1 254.4 232.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
SOHO0 HSO AH for SO3958 KJmol AH for HO2858 KJmol ...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
Given that the H f of 1 M H 2 SO 3 is -633 kJ, use the data in Appendix II, Table B to calculate the H for the formation of a 1 M solution of SO 2 in water from SO 2 (g). B. Standard Thermodynamic...
-
In the following exercises, you will use the data in the Solmaris Condominium Group database shown in Figures 1-21 through 1-25. (If you use a computer to complete these exercises, use a copy of the...
-
In Exercises evaluate the second derivative of the function at the given point. Use a computer algebra system to verify your result. (x) = cos x, (0, 1)
-
Develop brief answers to each of the following questions: 1. What is one way a company can improve its earnings per shares without improving its earnings or net income? 2. Why is comprehensive income...
-
The Butler-Huron Companys balance sheet and income statement for last year are as follows: *Assume that all sales are credit sales and that average accounts receivable are the same as ending accounts...
-
Problem 8 is the same as problem 7 with respect to initial measurement of the ARO liability. Now assume that Tadpoles credit standing improves over time, causing the credit-adjusted risk-free rate to...
-
The PENTEL Corporation manufactures three different types of computer chips. Each type of chip requires different amounts of processing time in three different departments as summarized in the...
-
(5 pts) In early 2020, The Week reported that the median wage for Costco's employees is almost $39,000. This is higher than the median wage for all Americans and much higher than Walmart's median...
-
Calculate the standard enthalpy of reaction for reducing the different forms of iron oxide to iron metal and CO 2 from the reaction of the oxide with CO. Identify which reaction is the most...
-
Breathing air that contains 0.13% CO by volume for 30 minutes will cause death. CO can form by incomplete combustion of carbon-containing compounds. Calculate the minimum volume of octane (C 8 H 18 ,...
-
2014 housing market data from37 ZIP Codes in Austin, Texas, is available online. Using the data file 2014_Housing_Market_Analysis_Data_by_ Zip_Code, and with the assistance of statistical software,...
-
In the event an employee retires, she will be paid for the 3/4 of her accumulated sick leave . If the employee has accumulated one hundred twenty-eight hours sick leave, how many day's sick leave...
-
James uses his automobile 80% for his business that he operates as a sole proprietorship. He drove a total of 14,000 miles and paid the following amounts for his vehicle: Parking at client's offices...
-
If a hospital makes $25,000,000 and has a 2.5% penalty, what is the reduction amount? (Assume the 2.5% penalty is the only penalty being assessed on the $25,000,000 in billings.)
-
4. You are working as an intern in an FBI digital forensics lab. Your job is to copy storage media and make the appropriate copies of that media for safekeeping and analysis. a. How many copies are...
-
Mulberry Company's first quarter budget reflects that direct materials are budgeted at $180,000, direct labor is budgeted at $360,000, and overhead is budgeted at $420,000. At the end of the first...
-
Emergency Economic Stabilization Act of 2008 included modifications to Form 1099-B. These changes imposed additional reporting criteria for custodians and brokers beginning in 2011 with equities,...
-
Troy is a qualified radiologist who operates a successful radiology practice from purpose- built rooms attached to his house. Troy works in the practice three days a week, and the other two days he...
-
Figure P20.43 shows two cases in which a positively charged particle is moving near a bar magnet. What is the direction of the magnetic force in each case? Case 2 Case 1 +9 +4
-
Figure P20.44 shows an electron moving near a current loop which lies in the xy plane with its center at the origin. If the electrons velocity is parallel to the y axis, what is the direction of the...
-
A long, straight wire of length 1.4 m carries current I = 3.5A. If a magnetic field of magnitude B = 1.5 T is directed perpendicular to the wire, what is the magnitude of the force on the wire?
-
Step One: Find two (2) scholarly journal articles published in 2022 on one of the topics covered in this course. You may define the topic as narrowly as you wish, but it should have some connection...
-
Bahama Bob has a sole proprietorship which operates a food truck preparing lunch and dinner items featuring the cuisine of the Bahamas. Bob parks the truck outside the Hospital of the University of...
-
Practice on-going and continuous quality improvement in maximizing competitiveness in order to produce goods or services of world-class quality Assignment description: This assignment requires a...

Study smarter with the SolutionInn App


