What is the net ionic equation for the reaction that occurs when aqueous solutions of KHCO 3
Question:
What is the net ionic equation for the reaction that occurs when aqueous solutions of KHCO3 and HBr are mixed?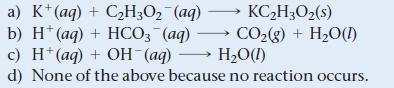
Transcribed Image Text:
KC₂H3O₂ (s) a) K+ (aq) + C₂H3O₂ (aq) b) H+ (aq) + HCO3 (aq) - c) H+ (aq) + OH-(aq) CO₂(g) + H₂O(1) H₂O(1) d) None of the above because no reaction occurs.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
b H ...View the full answer

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
412+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and HNO 3 are mixed? a) K+ (aq) + NO3 (aq) b) NO (aq) + OH(aq) c) H(aq) + OH(aq) HO(1) d) None of the above...
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and SrCl 2 are mixed? a) K (aq) + CI (aq) KCI(s) b) Sr+ (aq) + 2 OH-(aq) Sr(OH)2 (s) c) H* (aq) + OH (aq) ...
-
What is the net ionic equation for the reaction that occurs when an aqueous solution of KI is added to an aqueous solution of Pb(NO 3 ) 2 ?
-
Which statement about the pass-by-reference is NOT true? a.Every time you pass a reference variable to a method, you also pass the object referred by the refenerece variable to the method. b.In the...
-
What non-financial information would be important for an AIS to capture about a manufacturing firms production process?
-
During the compression stroke of a certain gasoline engine, the pressure increases from 1.00 atm to 20.0 atm. If the process is adiabatic and the fuelair mixture behaves as a diatomic ideal gas, (a)...
-
Show that A* is optimal and complete in most circumstances.
-
The Metropolitan Housing Authority is charged with providing housing to low-income residents. The authority is currently assessing two options, each of which would provide homes for 100 families....
-
Juan, priest of the San Juan Bautista Church in Culebra, is interested in purchasing a bus with a capacity of 12 passengers or more. When visiting your office, Juan has the idea of using the vehicle...
-
How can you predict whether a precipitation reaction will occur upon mixing two aqueous solutions?
-
A 10.0 mL sample of 0.20 M HBr solution is titrated with 0.10 M NaOH. What volume of NaOH is required to reach the equivalence point? (a) 10.0 mL (b) 20.0 mL (c) 40.0 mL
-
As of January 1, 2016, Shundra Inc. had a balance of $4,500 in Cash, $2,500 in Common Stock, and $2,000 in Retained Earnings. These were the only accounts with balances in the ledger on January 1,...
-
1. The product I have chosen is the Dell Laptop. Dell is a company that was founded in Texas and is currently head-quartered in Round Rock, Texas. They specialize in the technology industry and are...
-
Golden State Elixirs' tinctures are produced in four steps: extraction, blending, testing, and bottling. The first step is the extraction of cannabis oil from cannabis buds, also called cannabis...
-
Assuming a 12% annual interest rate, determine the present value of a five-period annual annuity of $6.200 under each of the following situations: Note: Use tables, Excel, or a financial calculator....
-
Data for Hermann Corporation are shown below: Selling price Variable expenses Contribution margin Per Unit Percent of Sales $ 110 77 100% 70 $ 33 30% Fixed expenses are $82,000 per month and the...
-
A ball is thrown straight up . It passes a 2.13 - m - high window 7.80 m off the ground on its path up and takes 1 . 3 0 s to go past the window. What was the balls initial velocity?
-
a. Identify major determinants of PB and PE ratios. b. How can the analyst use jointly the values of PB and PE ratios in assessing the merits of a particular stock investment?
-
Vince, Inc. has developed and patented a new laser disc reading device that will be marketed internationally. Which of the following factors should Vince consider in pricing the device? I. Quality of...
-
Consider the pulse described in terms of its displacement at t = 0 by where C is a constant. Draw the wave profile. Write an expression for the wave, having a speed v in the negative x-direction, as...
-
Write the expression for the wavefunction of a harmonic wave of amplitude 10 3 V/m, period 2.2 10 -15 s, and speed 3 10 8 m/s. The wave is propagating in the negative x-direction and has a value of...
-
Show that if the displacement of the string in Fig. 2.12 is given by y(x, t) = A sin [kx - Ït + ε] then the hand generating the wave must be moving vertically in simple harmonic...
-
Fill in the missing data for each of the following independent cases. (Ignore income taxes.) Note: Do not round intermediate calculations. Leave no cells blank - be certain to enter "0" wherever...
-
Comparing Three Depreciation Methods Dexter Industries purchased packaging equipment on January 8 for $187,000. The equipment was expected to have a useful life of four years, or 5,200 operating...
-
Ayden's Toys, Incorporated, just purchased a $500,000 machine to produce toy cars. The machine will be fully depreciated by the straight-line method over its 7-year economic life. Each toy sells for...

Study smarter with the SolutionInn App


