What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and
Question:
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and SrCl2 are mixed?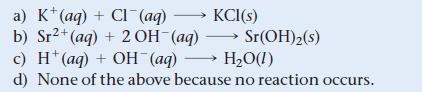
Transcribed Image Text:
a) K (aq) + CI (aq) →→→→KCI(s) b) Sr²+ (aq) + 2 OH-(aq) Sr(OH)2 (s) c) H* (aq) + OH (aq) →→→ H₂O(1) d) None of the above because no reaction occurs.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
2 b S...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and HNO 3 are mixed? a) K+ (aq) + NO3 (aq) b) NO (aq) + OH(aq) c) H(aq) + OH(aq) HO(1) d) None of the above...
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KHCO 3 and HBr are mixed? KCH3O (s) a) K+ (aq) + CH3O (aq) b) H+ (aq) + HCO3 (aq) - c) H+ (aq) + OH-(aq) CO(g) +...
-
What is the net ionic equation for the reaction that occurs when an aqueous solution of KI is added to an aqueous solution of Pb(NO 3 ) 2 ?
-
8. Be able to discuss how economic growth was differed across the three eras we discussed prior to WWI.
-
Search the web for unusual and interesting uses of RFID tags. Find at least two that are unusual and share those with your classmates.
-
Primara Corporation has a standard cost system in which it applies overhead to products based on the standard direct labor-hours allowed for the actual output of the period. Data concerning the most...
-
On June 14, 1988, Thomas John Heck Jr. executed a note promising to pay Paul D. Heck \($51,000\) at 7 percent interest compounded annually. The note contains the following payment terms: Perpetual 90...
-
a. Draw a network flow diagram for this problem. b. Create a spreadsheet model for this problem and solve it. c. What is the optimal solution? d. If H&J converted each non-US currency it owns...
-
Syarikat Penapisan Petroleum Berhad produces three joint products; Gasoline, Kerosene dan Diesel; and byproduct, i.e. Tar in the same refinery process. The split-off point incurred after the...
-
What are the solubility rules? How are they useful?
-
The presence of one of the ANSWER NOW! following ions within a compound indicates that a compound is soluble with no exceptions. Which ion? (a) OH (b) SO- (c) NO3
-
Outline the matters to be included in auditors communications with those charged with auditees governance.
-
Activities involved in the supplier relationship management (SRM) macro process include planning of internal production and storage. order fulfillment. supplier evaluation and selection. order...
-
Develop an optimization model to identify potential sites in a region.
-
The plant location model can also be modified to allow staff costs to be calculated. to allow direct shipments to the region. to allow direct shipments between factories and markets. to allow...
-
________ measures the incremental costs that are independent of the size of the order. Average sale price Incremental variable cost per unit Incremental fixed cost per order Incremental indirect cost...
-
Which of the following statements about the push processes is accurate? They may also be referred to as reactive processes. Execution is initiated in response to customer orders. At the time of...
-
On November 30, 2010, the Zu Company had the following account balances: During the month of December, the Zu Company entered into the following transactions: Date Transaction Dec. 4 Made cash sales...
-
The graph of the sequence whose general term is an = n - 1 is which of the following? [8.1] A. B. TITTT 3-2-1 23.45 2.3.4
-
Identify an actionreaction pair of forces in each of the following situations. (a) A person pushing on a wall (b) A book resting on a table (c) A hockey puck sliding across an icy surface (d) A car...
-
Figure P2.55 shows the position as a function of time for an apple that falls from a very tall tree.? (a) At what time does the apple hit the ground?? (b) Use a graphical approach to estimate and...
-
Figure P2.53 shows the position as a function of time for an object. (a) What is the average velocity during the period from t = 0.0 s to t = 10.0 s? (b) What is the average velocity between t = 0.0...
-
Explain the onchocerciasis control program: What year was it started? (1 pt) What control measures were first implemented and how? (2 pts) In what year did they start using the anthelminthic? (1 pt)...
-
Falling in line with their credit score, the estimated salary is provided whenever acustomer requests a new product. The number of products indicates the number of products eachcustomer uses, such as...
-
Naturally, the manager and staff did not receive a bonus. However, the manager was upset with this turn of events. "We all worked extra hard this year. It was a tough year. The fuel prices more than...

Study smarter with the SolutionInn App


