What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and
Question:
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and HNO3 are mixed?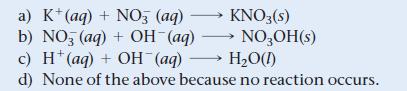
Transcribed Image Text:
a) K+ (aq) + NO3 (aq) b) NO (aq) + OH¯(aq) c) H(aq) + OH(aq) →→→ H₂O(1) d) None of the above because no reaction occurs. KNO3(s) NO3OH(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
c H...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and SrCl 2 are mixed? a) K (aq) + CI (aq) KCI(s) b) Sr+ (aq) + 2 OH-(aq) Sr(OH)2 (s) c) H* (aq) + OH (aq) ...
-
You are developing an accrual-adjusted income statement from a cash income statement. During the year, accounts payable increased. What is the adjustment you need to make?" add ending accounts...
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KHCO 3 and HBr are mixed? KCH3O (s) a) K+ (aq) + CH3O (aq) b) H+ (aq) + HCO3 (aq) - c) H+ (aq) + OH-(aq) CO(g) +...
-
1. Prepare program using threads in java that can print 10 times the numbers 1,2,3,4,5 in a series. 2. Prepare program using threads and a semaphore in java that can print the numbers 1,2,3,4,5 in a...
-
Figure is a system flowchart for P. Miesing and Companys purchase order event. Prepare a narrative to accompany the flowchart describing this purchase order event. Include in your narrative the...
-
Miller Toy Company manufactures a plastic swimming pool at its Westwood Plant. The plant has been experiencing problems as shown by its June contribution format income statement below: Contains...
-
On February 16, 2005, Joseph and Margaret Buset borrowed $192,000 from Fremont Investment & Loan (the Originator). The loan was secured by a mortgage on a residential condominium. The mortgage named...
-
Parnevik Company has the following securities in its investment portfolio on December 31, 2012 (all securities were purchased in 2012): (1) 3,000 shares of Anderson Co. common stock which cost...
-
The KLM Partnership, which uses the accrual method of accounting, is owned equally by Karen ( cash method taxpayer ) , and LM corporation ( accrual method taxpayer ) . Karen is a real estate...
-
Write complete ionic and net ionic equations for each reaction. (a) 3 SrCl (aq) + 2 Li3PO4(aq) + KOH(aq) (b) HCH3O2(aq) Sr3 (PO4)2(s) +6 LiCl(aq) HO(1) + KCH3O(aq)
-
Write an equation for the precipitation reaction that occurs (if any) when solutions of sodium nitrate and lithium sulfate are mixed.
-
In Exercises evaluate the limit, using LHpitals Rule if necessary. lim x1 In x sin TX
-
How can the full set of logistical and cross-functional drivers be used to create strategic fit for a furniture manufacturer, specializing in kitchen cabinetry, that targets both time-sensitive and...
-
What strategic options are considered by decision makers when designing a global supply chain network in the context of overcoming future uncertainties? Identify possible uncertainties that must be...
-
Which of these statements about Dells supply chain is most accurate? Dells supply chain surplus was largely driven by their negative shipping model. Dells initial success was largely driven by the...
-
Consider a retailer like Marks & Spencer (M&S), which is outsourcing parts of its delivery services to a logistics provider. When should such a company choose to operate its own account operation to...
-
Price decisions only affect buyers behavior and not the responsiveness of the supply chain. Comment on this statement with reference to shipping logistics.
-
Dorothy Corporation had the following accounts in its year-end adjusted trial balance: Accounts payable, $7,200; Salaries payable, $5,800; Income taxes payable, $4,000; Short-term notes payable,...
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
Argon-ion lasers typically generate multi-watt beams in the green or blue regions of the visible spectrum. Determine the frequency of such a 514.5-nm beam.
-
Consider the function where A is a constant. Show that it is a solution of the differential wave equation. Determine the speed of the wave and the direction of propagation. = (1 '2)p (z vt) + 1
-
Show that the function is a solution of the differential wave equation. In what direction does it travel? (y, t) = (y 41)
-
Circuit Masters Incorporated (CMI) is presently operating at 80% of capacity and manufacturing 128,000 units of a patented electronic component. The cost structure of the component is as follows: Raw...
-
Rentals Inc. leases a vehicle to United Inc. for four years on January 1 of Year 1, requiring equal annual payments on each January 1, with the first payment due at the lease commencement. The...
-
Hazelnut Corporation manufactures lawn ornaments. It currently has two product lines, the basic and the luxury. Hazelnut has a total of $137,912 in overhead. The company has identified the following...

Study smarter with the SolutionInn App


