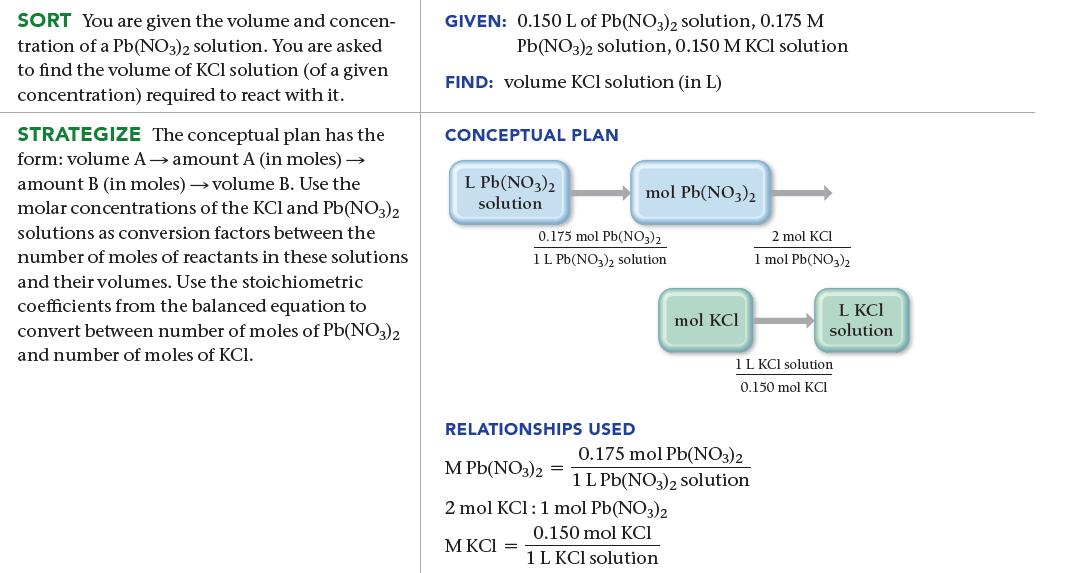
What volume (in L) of a 0.150 M KCl solution will completely react with 0.150 L of
Question:
What volume (in L) of a 0.150 M KCl solution will completely react with 0.150 L of a 0.175 M Pb(NO3)2 solution according to the following balanced chemical equation?
![]()
Transcribed Image Text:
2 KCl(aq) + Pb(NO3)2(aq) PbCl₂(s) + 2 KNO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
0150 L PbNO32 solution X 2 mol KCI 1 mol PbNO32 0175 mol PbNO32 1 LPbNO32 solution 1 L KCl solu...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Twelve years ago, Birch Ltd. (BL) borrowed $480,000 from Oak Trust Inc. (OTI). The 12- year, 10% note is due on todays date, December 31, 2020. The note was originally issued at par. BL is unable to...
-
What volume of 0.512 M NaOH will react with 17.9 g of H2C2O4(s) according to the following chemical equation? H2C2O4(s) + 2NaOH(aq) ( Na2C2O4(aq) + 2H2O()
-
In the Young's double slit experiment, interference fringes are formed using Na-light of 589 nm and 589.5 nm. Obtain the region on the screen where the fringe pattern will disappear. You may assume...
-
It's Yours Manufacturing wishes to maintain a growth rate of 8 percent a year, a debt-equity ratio of 0.45, and a dividend payout ratio of 25 percent. The ratio of total assets to sales is constant...
-
After plotting strategic risks and depicting the expected residual process risks after considering any control put in place to reduce the risks, what decision about designing audit tests can the...
-
The partial molal free energy of an element \(A\) in solution is the same as its (a) Chemical potential (b) Activity (c) Fugacity (d) Activity co-efficient.
-
On January 1, 2013, Plano Company acquired 8 percent (16,000 shares) of the outstanding voting shares of the Sumter Company for $192,000, an amount equal to Sumters underlying book and fair value....
-
How security officials determine vulnerabilities to natural, human-made, and technological threats. Also, listsocio-economic and criminal activity factors associated with the environment surrounding...
-
Analyze, Forecast, and Interpret Income Statement and Balance Sheet Following are the income statement and balance sheet of ADP Inc. Note: Complete the entire question using the following Excel...
-
Explain how a strong electrolyte, a weak electrolyte, and a nonelectrolyte differ.
-
The image shown at the far right represents a small ANSWER NOW! volume within 500 mL of aqueous ethanol (CH 3 CH 2 OH) solution. (The water molecules have been omitted for clarity.) Which of the...
-
1. Calculate and express as a percentage the companys debt-to-assets ratio using amounts reported in its financial statements for the years ended January 29, 2017, and January 31, 2016, respectively....
-
Billie owns a high-end home theater speaker system, which she bought 2 years ago for $2236. Unfortunately, the speaker system was stolen last week, but Billie has a renter's insurance. What is the...
-
What molecular mechanisms underlie the process of epigenetic regulation in cellular differentiation and development ?
-
Investigate the evolutionary origins and adaptive significance of complex biological phenomena, such as altruistic behavior, sociality, and cooperation, considering their genetic, ecological, and...
-
What are the uses or purpose of minutes of meeting? Other important information and the benefits of meeting minutes for work and business purposes. Distinguishes various business communication forms.
-
How do you differentiate between notice, agenda and minutes of the meeting? Describe the importance of minutes of meeting.
-
Why should public relations practitioners be concerned about employee speech?
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
Predict the major product(s) for each of the following reactions: (a) (b) (c) (d) (e) (f) CH NH2 [H*] (-H20) 1) PhMgBr 2) H20
-
Identify the starting materials needed to make each of the following acetals: (a) (b) (c) OEt
-
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
-
Read The Target Case A "Kill Chain" Analysis of the 2013 Target Data Breach. Define terms such as threats, safeguards and targets. What type of threats did Target face? Which specific techniques did...
-
Summerville is a lakeshore community in the Midwest. In its heyday, the town relied heavily on the automotive industry for its tax base and jobs for its residents. As the automotive industry...
-
How would you design an embedded system to measure the distance between an object and a sensor using ultrasonic waves? 2. Imagine you have to design an embedded system that monitors the health of an...

Study smarter with the SolutionInn App


