Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HI(aq) + RbOH(aq) b.
Question:
Write balanced complete ionic and net ionic equations for each acid–base reaction.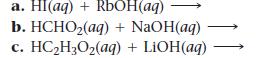
Transcribed Image Text:
a. HI(aq) + RbOH(aq) b. HCHO₂(aq) + NaOH(aq) c. HC₂H₂O₂(aq) + LiOH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The balanced complete ionic and net ionic equations f...View the full answer

Answered By

Larlyu mosoti
I am a professional writer willing to do several tasks free from plagiarism, grammatical errors and submit them in time. I love to do academic writing and client satisfaction is my priority. I am skilled in writing formats APA, MLA, Chicago, and Harvard I am a statistics scientist and I can help out in analyzing your data. I am okay with SPSS, EVIEWS, MS excel, and STATA data analyzing tools.
Statistical techniques: I can do linear regression, time series analysis, logistic regression, and some basic statistical calculations like probability distributions. . I'm ready for your working projects!
Services I would offer:
• Academic writing.
• Article writing.
• Data entry.
• PDF conversion.
• Word conversion
• Proofreading.
• Rewriting.
• Data analyzing.
The best reason to hire me:
- Professional and Unique work in writing.
- 100% satisfaction Guaranteed
- within required time Express delivery
- My work is plagiarism Free
- Great communication
My passion is to write vibrantly with dedication. I am loyal and confident to give my support to every client. Because Client satisfaction is much more important to me than the payment amount. A healthy client-contractor relationship benefits in the longer term. Simply inbox me if you want clean work.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write complete ionic and net ionic equations for each of the following molecular equations. a. 2HNO3(aq) + Mg(OH)2(s) 2H2O(l) + Mg(NO3)2(aq) Nitric acid, HNO3, is a strong electrolyte. b....
-
Write balanced complete ionic and net ionic equations for each reaction. a. HCl(aq) + LiOH(aq) HO(1)+ LiCI(aq) b. Cas(aq) + CuCl(aq) CuS(s) + CaCl(aq) c. NaOH(aq) + HCHO(aq) d. Na3PO4(aq) + NiCl(aq)...
-
Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HBr(aq) + NaOH(aq) b. HF(aq) + NaOH(aq) C. HCHO(aq) + RbOH(aq)
-
2. National Defense (40 points). There are 11 countries in Europe who get utility from general consumption c, and from European national defense G. The utility of a generic country i is u(ci, G) =...
-
Why are competent employees important to an organizations internal control system?
-
Using the formulas provided for utilization calculate the utilization of your classroom.
-
Presented here are the accounts of Plantscapes Decor Services Corporation for the year ended December 31, 2011. Requirements 1. Prepare Plantscapes Decor Services Corporations income statement. 2....
-
Use the data in problem 8 to create a Pareto diagram. Problem 8 In an apparel factory, every time a sewing machine breaks, the symptom is recorded. In the past 30 days, all of the sewing machine...
-
Examine the financial data in the most recent Annual Reports of various Australian supermarkets. Compare and contrast the results from each Supermarket. Which company is performing the best?...
-
A 25.00-mL sample of an unknown HClO 4 solution requires titration with 22.62 mL of 0.2000 M NaOH to reach the equivalence point. What is the concentration of the unknown HClO 4 solution? The...
-
Complete and balance each acidbase equation. a. HI(aq) + LiOH(aq) b. HCHO(aq) + Ca(OH)(aq) c. HCl (aq) + Ba(OH)(aq)
-
What is the beta of the following? a. Risk-free asset b. Market portfolio
-
(10 points) Marketing Message: Describe the message you plan to communicate to the target market. This should be a focused message that explains the benefits of the product/event in a way that...
-
Bidding-your-valuation is the unique symmetric BNE of the second-price auction (SPA) when bidders have SIPV. However, this is not the unique BNE in a SPA. In fact, there are other asymmetric...
-
y = x ^ 2 - 3 x + 4 x + y = 4 Solve the system of equations. Show your work.
-
Face value : 1000 $ Coupon rate : 10% (paid semi-annually) Yield : 9% (compound semi-annually) Maturity: 10 years Conversion ratio : 30 Price of shares : 41 $ Price of the bond ?
-
June 1 The owner, 3 4 4 5 7 7 8 11 12 _invested P 150,000. Purchased furniture P 29,000 plus 12% VAT for cash. Purchased goods on account from ABC Co., P 19,700 plus 12% VAT terms: 2/10, n/30. Paid...
-
Assume that the Clampton Company in the previous problem expects to pay income taxes of 40 percent and that a loss on the sale or disposal of equipment is treated as an ordinary deduction, resulting...
-
Explain the operation of the dividends received deduction.
-
An object is dropped from a height of 53 in. Neglecting air resistance, how long would it take for the body to strike the ground? Use a = g = 32.2 ft/s 2 . A body starting from rest with constant...
-
Calculate the kinetic energy in Nm of a 15-kg mass if it has a velocity of 1.20 m/s. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the kinetic energy in Nm of a 3600-kg truck moving at 16 km/h. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
A large portion of our working lives is spent working in Teams. How we communicate with our team members is crucial to the success (or failure) of the team. In this week's lesson, we looked at the...
-
summarize "What is Strategy" after watching Michael Porter's one-minute video on "What is Strategy" (link in Module 1) and reading Porter's "What is Strategy?" in Harvard Business Review 74, Issue 6,...
-
If you were appointed CM consultant how would you work with CEO and middle management to address the challenges & issues coming out of the adoption & move to AI at the company, Also, develop a step...

Study smarter with the SolutionInn App


