Without using Fig. 3.4, predict the order of increasing electronegativity in each of the following groups of
Question:
Without using Fig. 3.4, predict the order of increasing electronegativity in each of the following groups of elements.
Data in Fig. 3.4
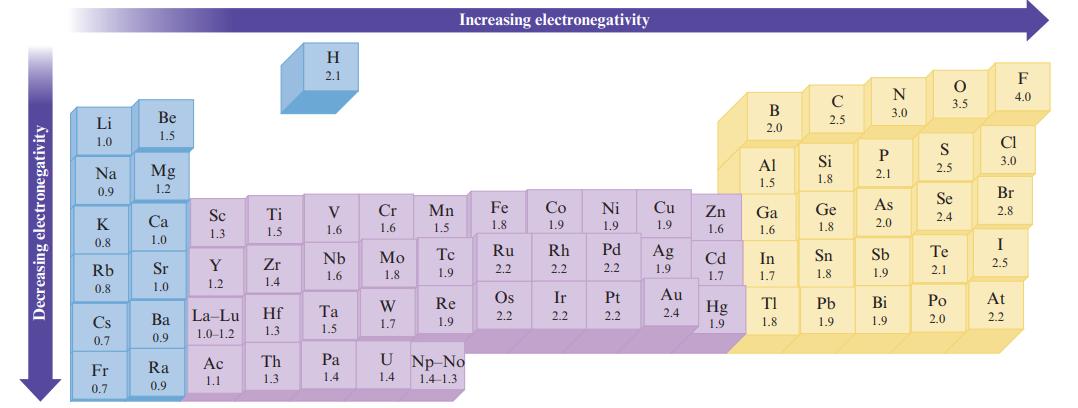
a. Na, K, Rb
b. B, O, Ga
c. F, Cl, Br
d. S, O, F
Transcribed Image Text:
Decreasing electronegativity Li 1.0 Na 8 0.9 K 0.8 Rb 0.8 Cs 0.7 Fr 0.7 Be 1.5 Mg 1.2 Ca 1.0 Sr 1.0 Ba 0.9 Ra 0.9 Sc 1.3 Y 1.2 La-Lu 1.0-1.2 Ac 1.1 Ti 1.5 Zr 1.4 Hf 1.3 Th 1.3 H 2.1 V 1.6 Nb 1.6 Ta 1.5 Pa 1.4 Cr 1.6 Mo 1.8 W 1.7 Mn 1.5 Tc 1.9 Re 1.9 Increasing electronegativity U Np No 1.4 1.4-1.3 Fe 1.8 Ru 2.2 Os 2.2 Co 1.9 Rh 2.2 Ir 2.2 Ni 1.9 Pd 2.2 Pt 2.2 Cu 1.9 Ag 1.9 Au 2.4 Zn 1.6 Cd 1.7 Hg 1.9 B 2.0 Al 1.5 Ga 1.6 In 1.7 ΤΙ 1.8 C 2.5 Si 1.8 Ge 1.8 Sn 1.8 Pb 1.9 P 2.1 N 3.0 As 2.0 Sb 1.9 Bi 1.9 S 2.5 O 3.5 Se 2.4 Te 2.1 Po 2.0 Cl 3.0 F 4.0 Br 2.8 I 2.5 At 2.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Without looking at Fig 34 Id predict the order of increas...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Without using Fig. 13.3, predict the order of increasing electronegativity in each of the following groups of elements. a. C, N, O b. S, Se, Cl c. Si, Ge, Sn d. Tl, S, Ge e. Na, K, Rb f. B, O, Ga...
-
Without using Fig. 3.4, predict which bond in each of the following groups will be the most polar. Fig. 3.4 a. C-H, Si-H, Sn-H b. Al-Br, Ga-Br, In-Br, Tl-Br c. C-O or Si-O d. O-F or O-CI Decreasing...
-
Without using Fig. 3.4, predict which bond in each of the fol- lowing groups will be the most polar. Data in Fig. 3.4 a. C-F, Si-F, Ge-F b. P-Cl or S-Cl c. S-F, S-Cl, S-Br d. Ti-Cl, Si-Cl, Ge-Cl...
-
Capital Inc. has prepared the operating budget for the first quarter of 2015. They forecast sales of $50,000 in January, $60,000 in February, and $70,000 in March. Variable and fixed expenses are as...
-
Why does a monopolist not charge the same price for the same good in two different countries?
-
The president of DePaul University wants to conduct a survey to determine the communitys opinion regarding campus safety. The president divides the DePaul community into three groups: resident...
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
The Bartram-Pulley Company (BPC) must decide between two mutually exclusive investment projects. Each project costs $6,750 and has an expected life of 3 years. Annual net cash flows from each project...
-
Our company makes school buses. Here is some data on the cost of producing these vehicles. Monthly Production Data Output 0 1 2 3 4 5 6 7 8 9 10 Total cost 200,000 400,000 575,000 725,000 850,000...
-
Describe the type of bonding that exists in the Cl 2 (g) molecule. How does this type of bonding differ from that found in the HCl(g) molecule? How is it similar?
-
On March 7, 2007, Toby Young was participating in a five-day cross-country ski and yoga program with Elderhostel and the Craftsbury Center, Inc. Young spent the afternoon cross-country skiing. A...
-
The lengths (in inches) of 150 newborn babies were recorded. Use whichever graphical technique you judge suitable to describe these data. What have you learned from the graph?
-
For the system given below, it is desired to adjust the opening and closing of the valve by generating a control input u to adjust the height (level) to ho, when h is the actual height. The total...
-
Describe the mechanisms employed by modern operating systems to manage security, including user authentication, access control lists (ACLs), and encryption. How do these mechanisms mitigate potential...
-
Given the following information, calculate the maintained markup %: opening inventory gross purchases Cost $69,600.00 Retail $145,000.00 returns to vendors net purchases freight $36,000.00 $75,000.00...
-
Hale Instruments, Inc., a C corporation, transferred its inventory to Agnes Acoustics, Inc., an S corporation, in a nonrecognition transaction. Agnes Acoustics took Hale Instruments' basis in the...
-
Audit risks for particular accounts and disclosures can be conceptualized in a model such that Audit risk (AR) = Inherent risk (IR) x Internal control risk (CR) x Detection risk (DR). Using this...
-
Throughout this text, we have many assignments based on the financial statements of Home Depot, Inc., in Appendix A. Refer to the financial statements to respond to the following items: a. Does the...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
When Na 3 PO 4 (aq) is added to a solution containing a metal ion and a precipitate forms, the precipitate generally could be one of two possibilities. What are the two possibilities?
-
Sodium chloride is listed in the solubility rules as a soluble compound. Therefore, the K sp value for NaCl is infinite. Is this statement true or false? Explain.
-
When 5 M ammonia is added to a solution containing Cu(OH) 2 (s), the precipitate will eventually dissolve in solution. Why? If 5 M HNO 3 is then added, the Cu(OH) 2 precipitate re-forms. Why? In...
-
Isabelle's has preferences represented by the Cobb Douglas utility function U(x1,x2)=x12 X2 with MRS = X1 She is currently consuming X1 3, X2 12. How many units of X2 will she be willing to give up...
-
[The following information applies to the questions displayed below.] A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term government and...
-
2. Alan Shepard, an all around badass human being, snuck a 6-iron golf club on board Apollo 14 and famously hit a golf ball on the moon. What a cool dude. The acceleration of gravity on the moon is...

Study smarter with the SolutionInn App


