Without using Fig. 3.4, predict which bond in each of the fol- lowing groups will be the
Question:
Without using Fig. 3.4, predict which bond in each of the fol- lowing groups will be the most polar.
Data in Fig. 3.4
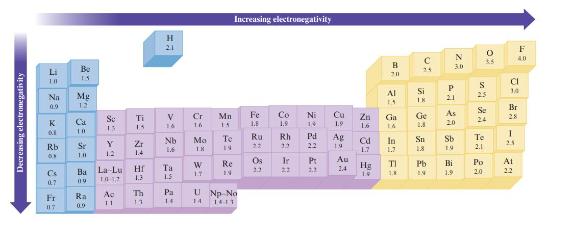
a. C-F, Si-F, Ge-F
b. P-Cl or S-Cl
c. S-F, S-Cl, S-Br
d. Ti-Cl, Si-Cl, Ge-Cl
Transcribed Image Text:
Decreasing electronegativity Li 18 K Na Mg 0.9 12 X 0. Cs 07 Be 20 Fr 117 Rb Se Y 1.2 de ²2:29 Ca 10 1.0 Ba 019 Ra Sc 13 0.9 Ti 15 Ac 11 Zr 1.4 La-Lut 10:12 1.3 = Th 14 H 21 V Nb 23 21 4: Hf Ta Pa 14 Cr 16 Mo IX W e p 1.7. Mn 14 To 19 Increasing electronegativity Re IN U Np No 14 1411 Fe 1.8 Ru 2# 8A 22 Os Co Ni 1.9 2:23 E Ir 22 14 Rh Pd 33 Pt 2.3 Cu 19 Ag 19 Au 24 398 25 1.6 Cd 1.7 B Zn Ga 14 Hg F 201 28 F3 TI с Si IK 33 1.8 Pb 30 27 29 82 N 21 As Sn Sb 18 20 19 Bi 1.9 0 S 25 Te 2.1 35 Se 24 Po 20 Cl 34 F 40 Br 28 -3 25. At 22
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
To predict which bond in each of the following groups will be the most polar we can use the followin...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Without using Fig. 3.4, predict which bond in each of the following groups will be the most polar. Fig. 3.4 a. C-H, Si-H, Sn-H b. Al-Br, Ga-Br, In-Br, Tl-Br c. C-O or Si-O d. O-F or O-CI Decreasing...
-
Without using Fig. 13.3, predict which bond in each of the following groups is the most polar. a. COF, SiOF, GeOF b. POCl, SOCl c. SOF, SOCl, SOBr d. TiOCl, SiOCl, GeOCl e. COH, SiOH, SnOH f. AlOBr,...
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
The following data applies to the two unrelated companies Lloyd Ltd and Cole Ltd: All taxable and deductible temporary differences relate to the profit or loss. Assume a corporate tax rate of 30%. A....
-
Given that real exchange rates fluctuate, when would be the best time to enter the market of a foreign country as an exporter to that market?
-
A sociologist wants to gather data regarding household income within the city of Boston. Obtain a sample using cluster sampling. Approach The city of Boston can be set up so that each city block is a...
-
In 2014, Javier Ramos decided to open seafood restaurants in California and Texas. Ramos claims that he was describing the crusted glaze applied to cooked seafood to his friend Ivan Murillo when...
-
Perfectionist Construction Company was the low bidder on an office building construction contract. The contract bid was $9,000,000, with an estimated cost to complete the project of $7,000,000. The...
-
What are some key factors in the development of the modern hospital in the United States? Is there one factor that stands out among the others through its importance?
-
Which of the following incorrectly shows the bond polarity? Show the correct bond polarity for those that are incorrect.
-
Describe the type of bonding that exists in the Cl 2 (g) molecule. How does this type of bonding differ from that found in the HCl(g) molecule? How is it similar?
-
Suppose that the economy is characterized by the following behavioral equations: Solve for the following variables. a. Equilibrium GDP (Y) b. Disposable income (Y D ) c. Consumption spending (C) C...
-
Suppose the supply and demand for a certain videotape are given by: 2; demand: p = - =q2 12 +30 supply: p = 59 where p is price and q is quantity. Find the equilibrium price.
-
A company is considering bidding for the exclusive rights to undertake a project, which will initially cost $70mln The company has forecast the following end of year cash flows for the four-year...
-
How much are you willing to pay for one share of Jumbo Trout stock if the company just paid a $0.70 annual dividend, the dividends increase by 2.5 percent annually, and you require a 10 percent rate...
-
ABC Corp wants to buy a machine to produce umbrellas. The machine costs $130,000 and will cost $7,500 to install. Once in place, the machine will produce an income of $25,000 every year. ABC Corp...
-
Suppose that the worker Dolores Fuertes (Second Officer) receives a notification of termination of her contract for work or service. (Seniority - January 1 2019), having received the notification of...
-
Environmental Services, Inc. performs various tests on wells and septic systems. A few of the companys business transactions occurring during August are described below: On August 1, the company...
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
If a solution contains either Pb 2+ (aq) or Ag + (aq), how can temperature be manipulated to help identify the ion in solution?
-
Silver chloride dissolves readily in 2 M NH 3 but is quite insoluble in 2 M NH 4 NO 3 . Explain.
-
The stepwise formation constants for a complex ion usually have values much greater than 1. What is the significance of this?
-
Carma Corp. currently pays a dividend of $2.00 per share. In addition, Carmas market beta is 2.0 when the risk free rate is 10% and the expected market risk premium is 5%. Estimate the intrinsic...
-
You are a manager at Northern Fibre, which is considering expanding its operations in synthetic fibre manufacturing. Your boss comes into your office, drops a consultant's report on your desk, and...
-
A 3.85 kg box is suspended from a system of massless wires attached to a ceiling. The first wire makes an angle of 01 with the ceiling, whereas the second wire makes an angle of 02 = 36.3" with the...

Study smarter with the SolutionInn App


