Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation,
Question:
Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation, acid–base, or gas-forming reaction. Show states for the products (s, ℓ, g, aq), give their names, and write the net ionic equation.
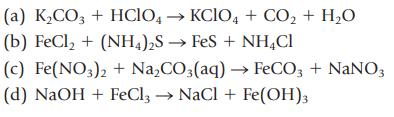
Transcribed Image Text:
(a) K₂CO3 + HCIO4 →KCIO4 + CO₂ + H₂O (b) FeCl, + (NH4)2S → FeS + NHẠCH (c) Fe(NO3)2 + Na₂CO3(aq) → FeCO3 + NaNO3 (d) NaOH + FeCl3 → NaCl + Fe(OH)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a KCO HClO KClO CO HO First balance the equation 2 KCO 2 HClO 2 KClO 2 CO 2 HO Now lets classify it ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (a) Cr2 (SO4)3(aq) + (NH4)2CO3(aq) (b) Ba...
-
The converter loop is a net generator of energy (why?) and proper utilization of that energy improves process economics. Assuming that the makeup gas is at 100C and that the purge gas and crude...
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. CrO3 + Ni2+ ( Cr3+ + Ni3+ b....
-
Why would an organization use outplacement strategies before downsizing? Outplacing employees helps former employees develop new skill sets. Outplacement strategies are a means of eliminating problem...
-
Can you suggest an event that would cause a countrys nominal interest rate to rise and its currency to appreciate simultaneously, in a world of perfectly flexible prices?
-
In this section we used a two step procedure to select a simple random sample of 30 EAI employees. Use this procedure to select a simple random sample of 50 EAI employees?
-
If a nurse disagrees with a physicians written orders, discuss what action the nurse should take to protect the patients safety.
-
The following regression was estimated for 23 quarters between 2004 and 2011 to test the hypothesis that tire sales (T) depend on new automobile sales (A) and total miles driven (M). Standard errors...
-
Based upon your in-depth knowledge of Lululemon, you are to identify and describe your firm's most persuasive growth opportunity: Your growth opportunity may fall within 1 of the 3 common situations...
-
Identify the primary species (atoms, molecules, or ions) present in an aqueous solution of each of the following compounds. Decide which species are Brnsted acids or bases and whether they are strong...
-
Aqueous solutions of ammonium sulfide, (NH 4 ) 2 S, and Hg(NO 3 ) 2 react to produce HgS and NH 4 NO 3 . (a) Write the overall, balanced equation for the reaction. Indicate the state (s, aq) for each...
-
________ are the smallest unit of language that carry meaning. a. Lexicon b. Phonemes c. Morphemes d. Syntax
-
Determine the maximum load for a simply supported beam of 5 0 ?mm diameter and 6 0 0 mm span, centrally loaded, as the load cyclically varies from W to 3 W . \ sigma en = 3 5 0 ?MPa, \ sigma yp = ...
-
Q.6. (a). Alternative method A and B using different tooling setups may be employed to manufacture a component on a particular machine tool whose operating cost (including wages of operator) is Rs....
-
Stress-life (S-N) curve approach for un-notched specimen S 0 Log N Stress-life (S-N) curve can be drawn with ..... Un-notched specimens Notched specimens Maximum stress : max Stress range : Number of...
-
A) Consider the planar double-wedge scramjet inlet geometry presented in Figure Q3A. As Mach number is changed from 1 M 5 the flow will be characterized by a number of qualitatively different shock...
-
A pipe of external diameter D = 71.5 mm and wall thickness t = 5.00 mm is connected to a square tube of external side length s = 64.4 mm and wall thickness t2 = 6.00 mm. A pipe here refers to a...
-
Create a flowchart from the following sequence of activities: Begin. Flow to activity A. Flow to decision B. If Yes, flow to activity C. If No, flow to activity D. From C flow to activity E and to...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
A carbocation is resonance stabilized when it is adjacent to an oxygen atom: Such a carbocation is even more stable than a tertiary carbocation. Using this information, propose a mechanism for the...
-
One liter of fully oxygenated blood can carry 0.18 liters of O 2 measured at T = 298 K and P = 1.00 atm. Calculate the number of moles of O 2 carried per liter of blood. Hemoglobin, the oxygen...
-
Determine whether the pinacol rearrangement, shown in the previous problem, is a reduction, an oxidation, or neither. Resonance stabilized H,SO, eat
-
The effective change in velocity, Av, required to launch from Earth's surface to Low Earth Orbit (LEO) is about 9.5 km/s.' Let's use Tsiolkovsky's rocket equation to design a space launcher capable...
-
How can conflict resolution frameworks, such as the Thomas-Kilmann Conflict Mode Instrument or the Dual Concern Model, be adapted and customized to suit the unique dynamics of specific industries or...
-
SpaceX will launch a new rocket. Ten engineers that developed the rocket were asked what they think the probability of successfully launching the rocket is. Here's the probability distribution for...

Study smarter with the SolutionInn App


