Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) SF4(g)
Question:
Balance the following equations, and name each reactant and product: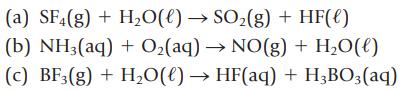
Data given in Example 3.1

Transcribed Image Text:
(a) SF4(g) + H₂O(l) → SO₂(g) + HF(e) (b) NH3(aq) + O₂(aq) → NO(g) + H₂O(l) (c) BF3(g) + H₂O(l) → HF(aq) + H₂BO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a SFg 2HOl SOg 4HFl Reactants Sulfur tetrafluoride gas and water Products ...View the full answer

Answered By

Amit Choudhary
I'm new in this profession regarding online teaching but previously i used to teach students near my college. I am teaching on online platform since last year and got good support from the students. I'm teaching on platforms like chegg and vedantu and also at my home in free time.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) FeO3(s) + Mg(s) MgO(s) + Fe(s) (b) AlCl3(s) + NaOH(aq) AI(OH)3 (s) + NaCl(aq) (c) NaNO3(s) +...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Which statement about the pass-by-reference is NOT true? a.Every time you pass a reference variable to a method, you also pass the object referred by the refenerece variable to the method. b.In the...
-
Describe the factors to consider before developing an aggregate plan.
-
The National Fire Protection Association reported that house fires in the United States result in $6.8 billion in losses annually (National Fire Protection Association, September 2015). Suppose a...
-
Water flows steadily from the pipe shown in Fig. P3.97 with negligible viscous effects. Determine the maximum flowrate if the water is not to flow from the open vertical tube at \(A\). Figure P3.97 3...
-
Cars A and B move in the same direction in adjacent lanes. The position x of car A is given in Figure, from time t = 0 to t = 7 .0 s. The figure's vertical scaling is set by xs = 32.0 m. At t = 0,...
-
Do you think that it would be more difficult to lead a non-profit group of employees versus a traditional organization? Why and why not? What type of leadership styles, skills, and qualities are...
-
Write balanced chemical equations for the following reactions. (a) The reaction of aluminum and iron(III) oxide to form iron and aluminum oxide (known as the thermite reaction). (b) The reaction of...
-
Balance the following equations: Data given in Example 3.1 (a) Cr(s) + Cl(g) CrCl3(s) (b) SiO (s) + C(s) Si(s) + CO(g) (c) Fe(s) + HO(g) Fe3O4(s) + H(g)
-
Mean and standard deviation. Write a program that reads a set of floating-point data values. Choose an appropriate mechanism for prompting for the end of the data set. When all values have been read,...
-
What is the entry for the current year's taxes on a common-sized income statement? Submit Answer format: Percentage Round to: 2 decimal places (Example: 9.24%, % sign required. Will accept decimal...
-
Investigate what historical events precipitated these ten amendments to the US Constitution. What were the fathers of the Constitution trying to protect and "clarify" with those 10 amendments?
-
2. (a) Explain five reasons why a marketing manager should understand consumer behaviour. (10 marks) (b) Fobel Limited segments its market psychographically. Explain five reasons that may have led...
-
Coasting Worldwide is a U . S . company headquartered in the state of New Jersey. The company provides chauffeured car services and limousines to executives of businesses located in New Jersey, New...
-
Prior to the Sarbanes - Oxley Act of 2 0 0 2 ( " SOX " ) , auditing companies had engaged in non - auditing functions with the corporations they audited. Although this had been cause for concern by...
-
What are joint costs? How do they affect the sell or process further decision?
-
For Problem estimate the change in y for the given change in x. y = f(x), f'(12) = 30, x increases from 12 to 12.2
-
Draw all significant resonance structures for each of the following compounds: a. b. c. d. e. f. g. h. i. j. k. l. . z: N.
-
A sealed flask with a capacity of 1.22 dm 3 contains 4.50 g of carbon dioxide. The flask is so weak that it will burst if the pressure exceeds 9.500 10 5 Pa. At what temperature will the pressure of...
-
A balloon filled with 11.50 L of Ar at 18.7C and one atm rises to a height in the atmosphere where the pressure is 207 Torr and the temperature is 32.4C. What is the final volume of the balloon?...
-
On June 30, 2021, Carnival sells a used computer (part of Property and Equipment) to a scrap dealer for $10 cash. The computer was purchased on June 30, 2020, for $100 and had an expected useful life...
-
The ice cream Company reported a pretax net operating loss of $300,000 for its year ended 20X1 for both financial reporting and for tax purposes. Ice cream will carry the operating loss forward. In...
-
For the equation, find three ordered pair solutions by completing the table. Then use any two of the ordered pairs to graph the equation. 1 y= 3x Complete the following table. y 0 C 10 8 6 4- 2 -10...

Study smarter with the SolutionInn App


