Benzene, C 6 H 6 , is a common compound, and it can be oxidized to give
Question:
Benzene, C6H6, is a common compound, and it can be oxidized to give maleic anhydride, C4H2O3, which is used in turn to make other important compounds.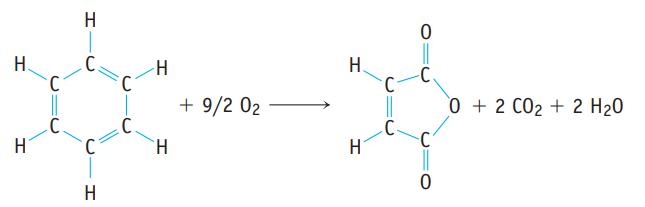
(a) What is the % atom economy for the synthesis of maleic anhydride from benzene by this reaction?
(b) If 972 g of maleic anhydride is produced from exactly 1.00 kg of benzene, what is the percent yield of the anhydride? What mass of the by-product CO2 is also produced?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





